Serviços Personalizados
Journal
Artigo
Links relacionados
Compartilhar
Odontoestomatología
versão impressa ISSN 0797-0374versão On-line ISSN 1688-9339
Odontoestomatología vol.23 no.38 Montevideo 2021 Epub 01-Dez-2021
https://doi.org/10.22592/ode2021n37e213
Research
Fluoride concentration in household salt marketed in Montevideo, Uruguay
1Departamento de Odontología Preventiva y Social, Facultad de Odontología, UFRGS, Porto Alegre, Brasil
2Cátedra de Odontopediatría, Facultad de Odontología, Universidad de la Republica, Montevideo, Uruguay
3Departamento de Odontología Preventiva y Social, Facultad de Odontología, UFRGS, Porto Alegre, Brasil. lhashizume@yahoo.com
Method:
Fourteen household salt brands marketed in Montevideo were analyzed to determine their free fluoride concentrations. Salt samples were weighed, diluted with deionized water, mixed with TISAB II, and F concentrations were determined using a fluoride-specific electrode connected to an ion analyzer.
Results:
Most salt brands showed values lower than 250 mgF/kg. Coarse salt samples showed higher F concentrations compared to the refined salt samples (p < 0.05). Salt brands with sodium fluoride had higher F concentrations than those with potassium fluoride (p < 0.05). Only two brands had F concentrations close to the content included in the product packages.
Conclusions
Household salt brands marketed in Montevideo (Uruguay) have significant variability in their F concentrations.
Keywords: fluoride; salt; dental caries; Uruguay
Objetivos:
Evaluar la concentración de fluoruro en la sal de mesa disponible comercialmente en Montevideo, Uruguay.
Método:
Fueron analizados catorce paquetes de sal para determinar la concentración de iones fluoruro libres. Las muestras se pesaron, se diluyeron en agua desionizada, se mezclaron con TISAB II y se utilizó un electrodo específico conectado a un analizador de iones para determinar la concentración de fluoruro
Resultado:
La mayoría de los paquetes presentaron valores inferiores a 250 mgF / kg. Las muestras de sal gruesa mostraron concentraciones mayores a las de sal fina (p <0.05). Aquellas conteniendo fluoruro de sodio presentaron mayor concentración de fluoruro que aquellas conteniendo fluoruro de potasio (p <0.05). Dos marcas presentaron concentraciones de fluoruro cercanas a las informadas en el envase.
Conclusiones:
Las sales de mesa comercializadas en Montevideo, Uruguay presentaron gran variabilidad en las concentraciones de fluoruros.
Palabras clave: fluoruros; sal; caries dental; Uruguay
Objetivos:
O objetivo deste estudo foi avaliar as concentrações de flúor (F) no sal de uso doméstico comercializado em Montevidéu, Uruguai.
Métodos:
Quatorze marcas de sal doméstico comercializadas em Montevidéu (Uruguai) foram analisadas para determinar as concentrações de F. As amostras de sal foram preparadas e as concentrações de flúor foram determinadas através de um eletrodo específico para F conectado a um analisador de íons.
Resultados:
A maioria das marcas de sal mostraram valores menores que 250mgF/kg. Amostras de sal grosso apresentaram as maiores concentrações de F comparadas às amostras de sal refinado (p < 0,05). Apenas duas marcas tiveram as concentrações de F próximas àquelas informadas nas embalagens dos produtos.
Conclusões:
As marcas de sal de uso doméstico comercializadas em Montevidéu (Uruguai) apresentam uma grande variabilidade em suas concentrações de F.
Palavras chave: fluoreto; sal; cárie dentária; Uruguai
Introduction
Community access to fluoride through water and salt has proven to be an effective public health measure to reduce the incidence of dental caries worldwide.1 Salt fluoridation has increased, mainly in developing countries, where water fluoridation has proven to be inaccessible due to decentralized and inadequate water supply and distribution systems.2-4
Community salt fluoridation has been available in Uruguay since 1991, reaching almost the entire population.5 Compared to water fluoridation, this alternative showed a similar reduction in caries prevalence while providing consumers the possibility to choose to buy the product.2 The Uruguayan population has access to fluoridated household salt at a 250 mgF/kg concentration.6
Studies conducted in other Latin American countries analyzed the fluoride concentrations of household salts and found significant variations between salt brands.7-12 In addition, most salt brands did not have the fluoride concentration reported on the product label, which goes against the country's legislation. However, no studies reporting the fluoride content of salt marketed in Uruguay were found. Therefore, this study aimed to evaluate the fluoride concentration of different brands of commercially available household salt in Montevideo, Uruguay.
Methodology
Sample
An analytical study was conducted to evaluate the fluoride concentration in fourteen fluoridated salt packages commercially available in supermarkets and grocery stores located in different areas of Montevideo, Uruguay. Three packages of the same brand but with different lot numbers were purchased. Samples were coded with different numbers to enable blind analysis. All the samples were analyzed between October 2016 and January 2017, before their expiration date. Table 1 shows the information reported on the product label provided by the manufacturer.
Fluoride concentration analysis
The salt samples were analyzed to determine their free fluoride concentrations. The analysis proceeded as follows: 0.025 g of salt was diluted in tubes containing 1 mL distilled and deionized water (w/v). Each sample was mixed in 1:1 (v/v) with TISAB II (Fisher Scientific Co., USA). Then the samples were analyzed with a fluoride-specific electrode connected to an ion analyzer, following Mier et al. (2009).13
Ten samples were analyzed in duplicate at two different times-at one-week intervals-to determine intra-examiner reproducibility. Additional analyses were performed to explain the differences observed in the initial results. Standard fluoride solutions ranging from 0.25 to 16.0 µg F/ml, mixed with TISAB II (1:1, v/v) added to 0.025 g NaCl/mL were used to calibrate the equipment (Orion 96-09 electrode coupled to an Orion Star A214 ion analyzer; Thermo Scientific, Cambridge, MA, USA). Each solution was mixed in 1:1 (v/v) in a flask with TISAB II.13
Table 1: Information provided by manufacturers on salt packages
| Brand name (Manufacturer) Country | Code | Salt type | Fluoride compound | Declared fluoride concentration (mg/kg) | Other active components |
| Monte Cudine (Haidar y cia. SRL) Argentina | 1 | Fine | NaF | 250 | NaCl; Potassium iodate; INS 341iii |
| Monte Cudine (Haidar y cia. SRL) Argentina | 2 | Coarse | NaF | 250 | NaCl; Potassium iodate; INS 341iii |
| Sal Sek (Deambrosi) Uruguay | 3 | Fine | NaF | 250 | Potassium iodate 30 ppm/Tricalcium phosphate 0.3% |
| Sal Sek (Deambrosi) Uruguay | 4 | Coarse | KF | 250 | Potassium iodate 30 ppm/Tricalcium phosphate 0.3% |
| Urusal (Anti SA) Uruguay | 5 | Coarse | NaF | 250 | NaCl; Potassium iodate 30 ppm |
| Urusal (Anti SA) Uruguay | 6 | Fine | NaF | 250 | NaCl; Potassium iodate 30 ppm/3413iii |
| Dos Anclas (Salinas Grandes, Hidalgo La Pampa) Argentina | 7 | Coarse | Not declared | 250 | 30 ppm iodine |
| Dos Anclas (Salinas Grandes, Hidalgo La Pampa) Argentina | 8 | Fine | Not declared | 250 | 30 ppm iodine/silicon dioxide 1.5% INS 515 |
| Celusal (Industrias químicas y mineras Timbo SA) Argentina | 9 | Coarse | KF | 250 | NaCl; Potassium iodate 30 ppm |
| Celusal (Industrias químicas y mineras Timbo SA) Argentina | 10 | Fine | KF | 250 | NaCl; Potassium iodate 30 ppm / INS 551ioins 536 |
| Cololo (Solsire SA) Uruguay | 11 | Coarse | KF | 250 | Potassium iodate 30 ppm |
| Cololo (Solsire SA) Uruguay | 12 | Fine | NaF | 250 | Potassium iodate/tricalcium phosphate |
| Dos Estrellas (Salinas Grandes, Hidalgo La Pampa) Argentina | 13 | Coarse | Not declared | 250 | 30 ppm iodine |
| Marina Diamante (Romani SA) Brazil | 14 | Fine | Not declared | 250 | NaCl, potassium iodate, anti-fogging, tricalcium phosphate, 30 ppm iodine |
NaF: sodium fluoride, KF: potassium fluoride
Data analysis
We performed a linear regression of the fluoride concentrations' logarithm of the standard solutions and calculated the respective mV values. The mathematical regression equation was used to determine the fluoride concentration of the salt solutions (in milligrams of fluoride). Mean concentrations and standard deviations were calculated for both aliquots in each flask and for the three flasks of each salt brand.
The Student's t-test was used to compare the salt groups (coarse vs. fine and NaF vs. KF). Statistical significance was set at p < 0.05. All statistical analyses were performed with SPSS version 18.0 (SPSS Inc., Chicago, IL). The concentrations found in the salt brands analyzed were compared to the information provided in the package label.
Results
Of the fourteen brands of fluoridated salt commercially available in Montevideo and analyzed in this study, only six were produced in Uruguay (samples 3, 4, 5, 6, 11, and 12). The other eight salt brands were imported from Argentina (samples 1, 2, 7, 8, 9, 10, and 13) and Brazil (sample 14).
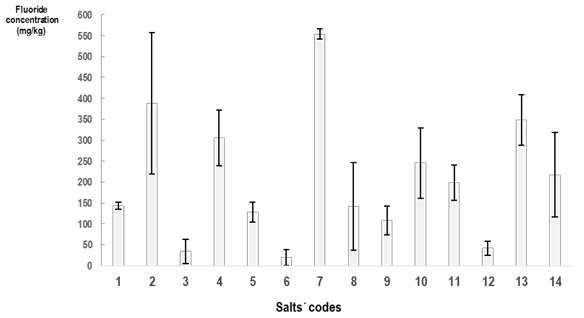
Figure 1 shows the mean fluoride concentration values found in each salt sample analyzed. They ranged from 19.22 to 553.42 mgF/kg. Most commercial brands available in Uruguay had fluoride concentration values below 250 mgF/kg. Only two salt brands (samples 10 and 14) reached the mean fluoride concentration expected (250 mgF/kg), and three brands had very high values (samples 2,4,7, and 13). A significant difference was detected when comparing the fluoride concentration values in different lots of the same salt brand, reflecting a wide standard deviation (Figure 1).
Six of the salt brands analyzed were available in two different presentations: fine and coarse. The mean value of fluoride concentration in the fine salts was 115.56 ± 109.09 mgF/kg (mean ± SD), with a coefficient of variation (CV) = 94.4%. The mean value of fluoride concentration in coarse salts was 242.33 ± 128.35 mgF/kg (mean ± SD), with a CV = 53.0%. In all salt brands, coarse salt presented higher fluoride concentration values than the fine salt concentration of the same brand (p < 0.05).
Sodium fluoride (NaF) and potassium fluoride (KF) were the compounds included in the salt brands analyzed. Salt brands containing KF (samples 4, 9, 10, and 11) had higher fluoride concentrations compared to salt brands containing NaF (samples 1, 2, 3, 5, 6, 12) (p < 0.05). The fluoride concentration of the salts with NaF was 125.76 ± 150.77 mgF/kg (mean ± SD), with a CV = 120%, while the salt brands with KF had a 214.49 ± 97.89 mgF/kg (mean ± SD) concentration, with a CV = 45.6%.
Discussion
In 1991, Uruguay introduced the nationwide National Salt Fluoridation Program at a concentration of 250 mgF/kg. In many Latin American countries, all salt for human consumption is fluoridated, while only household salt is fluoridated in Uruguay. As a result, specific legislation has been developed, and now the Uruguayan government requires 60% of household salt to be iodized and fluoridated throughout the country.6
Choosing a population-based approach to fluoride use benefits the general population, including people from lower socioeconomic status who lack access to fluoride toothpaste and professional fluoride applications. Several Latin American countries such as Colombia, Costa Rica, Mexico, and Jamaica use fluoridated salt as a population-based method to distribute fluoride. Uruguay is one of the countries that covers almost the entire population.14
The fluoride content of most salt brands available in Uruguay and analyzed in this study differed from the value declared on the package and that recommended by law. Even though the label indicates that the salt marketed has 250 mgF/kg, the values found in this study contradict this information. The fluoride concentration values of the samples analyzed ranged from very low-19.22 mgF/kg-to very high -553.42 mgF/kg. Only two brands had a mean content value close to the declared 250 mgF/kg (samples 10 and 14).
Several salt brands showed high standard deviations, reflecting the large difference in results found in different lots of the same brand (Figure 1). Therefore, it can be assumed that there is no adequate quality control in the salt fluoridation process that standardizes the amount to include in all the lots manufactured.
Also, fluoride is probably not added uniformly to the salt particles in each package. The size of the salt particles might affect the incorporation of fluoride. When comparing fine and coarse salt of the same brand, coarse salts had higher fluoride values than fine salts in all the analyzed samples.
The salt fluoridation method may also influence the results. The salt brands fluoridated with the wet method (samples 4, 9, 10, and 11), used KF as the fluorinated compound and had higher fluoride concentrations than those fluoridated with the dry method, using NaF (samples 1, 2, 3, 5, 6, 12). In addition, the salt brands treated with the wet method showed less variability in fluoride concentration than those treated with the dry method.
Two observational studies15,16 and one clinical trial(18) compared various methods of fluoride distribution at the population level (fluoridated salt and fluoridated water). The studies conducted by Mejia et al. (1976)17 and Sagheri et al. (2007)15 found no differences between fluoridation methods, which reinforces the critical role of fluoridated salt in areas where water fluoridation is not possible. Mejía et al. (1976)17 conducted their study in different Colombian communities, while Sagheri et al. (2007)15 conducted theirs in Dublin (fluoridated water) and Fribourg (fluoridated salt).
However, Fabruccini et al. (2016)16 reached different conclusions from the previous studies. They compared the effect of water fluoridation in Porto Alegre, Brazil, to salt fluoridation in Montevideo, Uruguay. The results showed that fluoridated salt provides less protection against dental caries in schoolchildren than fluoridated water. Additionally, children consuming fluoridated salt had a significantly higher DMFT than those exposed to fluoridated water, regardless of the criterion used (WHO or modified WHO). The authors attributed these results to the fact that the salt fluoridation program in Uruguay is limited to household salt and does not include eating facilities and restaurants, which may have reduced its impact on the population.16
This is the first study to analyze fluoride concentrations in the household salt brands available in Uruguay. This study showed that most Uruguayan fluoridated salt brands had fluoride concentrations lower than those recommended to be effective against dental caries. This may also help understand the results of Fabruccini et al. (2016),16 who found that the protective effect of fluoridated salt available in Uruguay was lower than that of fluoridated water from Brazil. The variability of fluoride concentration compared to package labels and recommendations suggests the need to improve the regulation of the manufacturing process. The Uruguayan population only has this community method to use fluoride as a public health measure to prevent dental caries. The results show that the population does not benefit from this method because it is exposed to either very low or very high fluoride levels.
Maupomé-Carvantes et al. (1995)8 also evaluated fluoride concentration in salt samples in Mexico City and found similar results. Of the 221 salt packages analyzed by the authors, the majority had a fluoride content below the official standard, as 50.2% of the samples had 1 to 50 mgF/kg of fluoride, while the package labels declared 250 mg F/kg.
For salt fluoridation in Uruguay to be effective at the population level and in controlling dental caries in epidemiological indices over the years, it is necessary to improve the salt manufacturing process with greater control during the household salt fluoridation stage. This would help all brands include the recommended fluoride values regardless of the lots manufactured, the type of fluoridation, and the salt size so consumers buy a quality product.
Although the results are based on the analysis of products marketed in 2016, they remain valid because the surveillance mechanisms of the National Salt Fluoridation Program have not changed.
Conclusion
There is significant variability in fluoride concentrations in commercially available brands of fluoridated household salt in Montevideo, Uruguay. Most of the salt brands analyzed do not include the concentration declared on the product label: the real contents range from very low to much higher than expected. The Uruguayan population might not be receiving the expected benefits from this method due to the lack of quality control, so it is necessary to better regulate the manufacturing process of fluoridated household salt.
REFERENCES
1. Tenuta LMA, Cury JA. Fluoride: its role in dentistry. Braz Oral Res. 2010; 24 (Suppl 1):9 -17. [ Links ]
2. Estupiñán-Day S. Promoting oral health: the use of salt fluoridation to prevent dental caries. Washington: PAHO, 2005. [ Links ]
3. Horowitz HS. Decision-making for national programs of community fluoride use. Community Dent Oral Epidemiol. 2000; 28(5):321-9. [ Links ]
4. Marthaler T, Petersen P. Salt fluoridation- an alternative in automatic prevention of dental caries. Int Dent J. 2005;55(6):351-8. [ Links ]
5. Ditterich RG, Rodrigues CK, Wambier DS. Fluoride salt as a alternative in public oral health: advantages and disadvantages. Rev Inst Ciênc Saúde, 2005; 23(3): 231-4 [ Links ]
6. Uruguay. Ministerio de Salud Pública. Comisión Honoraria de Salud Bucal. Encuesta de Salud Bucal en escolares de 11 a 14 años. Sector Público. República Oriental del Uruguay. 1999. [ Links ]
7. Martínez-Mier EA, Soto Rojas AE, Buckley CM, Stookey GK, Zero DT, Margineda J. Evaluación del contenido de flúor en la sal de mesa fluorada. Salud Pública Mex. 2004 May-Jun; 46(3):2003-4. [ Links ]
8. Walsh K I and Cury J A. Fluoride concentrations in salt marketed in Managua, Braz. Oral Res. 2018;32: e45 [ Links ]
9. Hernández-Guerrero JC, Fuente-Hernández J, Jiménez-Farfán MD, Ledesma-Montes C, Castañeda-Castaneira E, Molina-Frechero N, et al. Fluoride content in table salt distributed in Mexico City, Mexico. J Public Health Dent. 2008; 68(4):242-5. [ Links ]
10. Franco AM, Saldarriaga A, Gonzalez MC, Martignon S, Arbelaez MI, Ocampo A. Concentración de fluor en la sal de cocina en cuatro ciudades colombianas. Revista CES Odontología 2003;16: 21-6. [ Links ]
11. Tovar Valencia S, Castrillon Misnaza S. Perspectiva del uso del flúor vs caries y fluorosis dental. Colombia: Minsalud; 2016 [cited 2017 Sep 29] Disponible en: http://www.minsalud.gov.co/sites/rid/Lists/Bibliotecadigital/RIDE//VS/PP/ENT/perspectiva-uso-fluor.pdf. [ Links ]
12. Walsh KI, Cury JA. Fluoride concentrations in salt marketed in Managua, Nicaragua. Braz Oral Res. 2018; 24, 32:e45. [ Links ]
13. Mier EAM, Rojas AES, Buckley CM, Margineda J, Zero DT. Evaluation of the direct and diffusion methods for the determination of fluoride content in table salt. Community Dent Health, 2009; 26(4): 204-10. [ Links ]
14. Marthaler TM. Salt Fluoridation and Oral Health. Acta Medica Academica, 2013; 42(2): 140-55. [ Links ]
15. Sagheri D, Mcloughlin J, Clarckson J J. A comparison of dental caries levels in two communities with different oral health prevention strategies stratified in different social classes. Journal of Public Health Dentistry, 2007; 67(1): 1-7. [ Links ]
16. Fabruccini A, Alves LS, Alvarez L, Alvarez R, Susin C, Maltz M. Comparative Effectiveness Of Water And Salt Community-Based Fluoridation Methods In Preventing Dental Caries Among Schoolchildren. Community Dent. Oral Epidemiol, 2016; 44(6): 577-85. [ Links ]
17. Mejía R, Espinal F, Vélez H, Aguirre M. Fluoruración de la sal en cuatro comunidades colombianas. VIII. Resultados obtenidos de 1964 a 1972. Bol Oficina Sanit Panam. Colombia, 1976; 80: 205. [ Links ]
Conflict of interest: The authors declare that they have no conflict of interest regarding this study and reporting its results.
Authors' contribution note: 1.Conception and design of study 2.Acquisition of data 3.Data analysis 4.Discussion of results 5.Drafting of the manuscript 6.Approval of the final version of the manuscript. APDall’Onder has contributted in: 1,2,3,4,5,6 AFF has contributted in: 2,3,4,6 LAL has contributted in: 2, 3, 4,5,6 LNH has contributted in: 1,2,3,4,5,6
Received: February 24, 2021; Accepted: June 28, 2021










 texto em
texto em