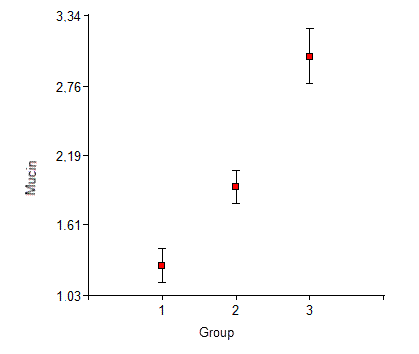Serviços Personalizados
Journal
Artigo
Links relacionados
Compartilhar
Odontoestomatología
versão impressa ISSN 0797-0374versão On-line ISSN 1688-9339
Odontoestomatología vol.23 no.38 Montevideo 2021 Epub 30-Set-2021
https://doi.org/10.22592/ode2021n37e206
Research
Salivary mucin concentration in patients with periodontal disease
1Cátedra Física Química Biológica -Facultad Odontología, Universidad Nacional del Nordeste, Argentina, odontoacuna@gmail.com
2Cátedra Fisiología Humana, Facultad Odontología, Universidad Nacional del Nordeste, Argentina
This work aimed to study the relationship between salivary mucin concentration and periodontal disease.
The sample was divided into three groups of 20 individuals each: Group 1 with no periodontal disease, Group 2 with gingivitis, and Group 3 with periodontitis.
Mucin was detected in all the saliva samples. Group 1 had an average value of 1.27 mg/ml. Group 2 had an average value of 1.93 mg/ml. Group 3 had an average value of 3.01 mg/ml.
The analysis of variance and subsequent F test (F = 25.01, p < 0.0001) confirmed significant differences in mucin content between the groups.
Increased salivary mucin concentration in periodontal patients could be a useful chemical marker for the clinical diagnosis of periodontal disease.
Keywords: saliva; mucin; gingivitis; periodontitis
El objetivo de este trabajo fue estudiar la relación entre la concentración de mucina salival y la enfermedad periodontal.
La muestra se dividió en tres grupos de 20 individuos cada uno: Grupo 1 sin enfermedad periodontal; Grupo 2 con gingivitis; y Grupo 3 con periodontitis.
En todas las muestras salivales se confirmó la presencia de mucina, el Grupo 1 presentó un valor promedio de 1,27 mg/ml. En el Grupo 2 se registró un promedio de 1,93 mg/ml. En el Grupo 3 se observó un promedio de 3,01 mg/ml.
El Análisis de la Variancia y posterior prueba de F (F = 25,01, p < 0,0001) confirman diferencias significativas en los contenidos de mucina entre grupos.
El aumento de la concentración de mucina salival en pacientes periodontales podría representar un marcador químico de utilidad como coadyuvante en el diagnóstico clínico de esta enfermedad.
Palabras claves: saliva; mucina; gingivitis; periodontitis
O objetivo deste trabalho foi estudar a relação entre a concentração de mucina salivar e a doença periodontal.
A amostra foi dividida em três grupos de 20 indivíduos cada: Grupo 1 sem doença periodontal; Grupo 2 com gengivite; e Grupo 3 com periodontite.
Em todas as amostras salivares foi confirmada a presença de mucina, o Grupo 1 apresentou valor médio de 1,27 mg / ml. No Grupo 2, foi registrada uma média de 1,93 mg / ml. No Grupo 3 foi observada uma média de 3,01 mg / ml.
A Análise de Variância e o teste F subsequente (F = 25,01, p <0,0001) confirmam diferenças significativas nos conteúdos de mucina entre os grupos.
O aumento da concentração de mucina salivar em pacientes periodontais pode representar um marcador químico útil como adjuvante no diagnóstico clínico desta doença.
Palavras-chave: saliva; mucina; gengivite; periodontite
Introduction
The term periodontal disease (PD) encompasses a broad group of infectious and inflammatory diseases, metabolic disorders and neoplasms that can affect the gingiva, periodontal connective tissues, and alveolar bone. Clinically, it can be classified into gingivitis (the lesion affects the gingiva) and periodontitis (the lesion affects the periodontal ligament, cementum, and alveolar bone) and it causes areas of local inflammation and tissue destruction 1. Epidemiologically, it is the second most prevalent oral cavity pathology worldwide 2. The presence of pathogenic bacteria in biofilm is one of the most predisposing factors for periodontal disease 3. At present, the diagnostic tools used to confirm or rule out this pathology only detect tissue destruction and do not determine if the disease is active or inactive. This makes it difficult for clinical dentists to treat patients correctly 4.
Saliva is an easy-to-access, non-invasive secretion. Taking a saliva sample for further analysis is not traumatic for the patient. Some elements present in saliva can be used as biochemical markers for periodontal disease. These can help determine if the pathology is active or inactive and if it is in its initial stage. These elements can include proteins, enzymes such as salivary alkaline phosphatase, salivary amylase, among others 5-6-7.
Another potential chemical marker is salivary mucin: a protein present in saliva that performs several functions in the oral cavity. For instance, it protects the body from invading microorganisms by binding to them and agglutinating them. Therefore, we believe that mucin concentration may be elevated in periodontal disease 8-9-10-11-12-13.
This work aimed to study the relationship between salivary mucin concentration and periodontal disease.
Materials and methods
This study included patients aged between 18 and 60 of both sexes who attended the School of Dentistry of Universidad Nacional del Nordeste (UNNE), Argentina. The patients were selected according to their periodontal disease diagnosis following specific clinical parameters (gingival index, bleeding on probing, pocket depth and attachment loss), radiographic findings and their will to participate in the study by signing an informed consent form. Patients excluded from the sample: pregnant women, patients previously treated for periodontal disease or with antibiotics or other medicines in the six months before the appointment, patients with chronic diseases such as diabetes or high blood pressure, psychological problems or impaired cognitive abilities, and patients who did not give their consent to participate in the study.
The patients underwent a clinical periodontal evaluation and were divided into three groups of 20 individuals: Group 1 (control group): patients with no periodontal disease; Group 2: patients with gingivitis; and Group 3: patients with periodontitis.
Saliva samples were taken to determine mucin concentration in each patient. All unstimulated saliva samples were collected between 8:00 a.m. and 10:00 a.m. 14
The saliva samples were collected in one session for each individual at a preset time, using disposable, sterile, screw-capped, 15-ml-propylene tubes, and then placed in test tubes. Then 1% Alcian blue in 50 ml of sodium acetate (buffer) was added and incubated at room temperature under continuous stirring for 30 minutes. After incubation, the samples were centrifuged for 20 minutes at 3000 rpm; 1 ml of 95% ethanol was added and shaken for 10 seconds. After five minutes, the samples were centrifuged for 20 min at 3000 rpm; then Aerosol OT was added (1:2 dilutions with distilled water) followed by an equal amount of ethyl ether with vigorous shaking. The samples were centrifuged for 15 minutes at 3000 rpm, and the optical density was measured at 605 nm using a UV spectrophotometer. The mucin concentration was calculated from this reading using a standard graph 15. All these measurements were taken at the Physicochemical Area of the Scientific Research Laboratory of the School of Dentistry, UNNE.
Patients’ gender (F and M) and age (in years) were also recorded. They were subsequently divided into three age categories: A1 - 18 to 33, A2 - 34 to 46, and A3 - 47 to 60.
The information was entered into an Excel spreadsheet and analyzed using InfoStat 2020 statistical software 16. An exploratory analysis of the data was conducted graphically and analytically to analyze the general behavior of the information. The associations between variables and their significance (p= 0.05) were studied with Pearson’s Correlation Coefficient (quantitative variables) and with independence tests using the Chi-square test (qualitative variables). Analysis of variance and F tests were performed to establish significant differences (p= 0.05) in mucin values between the different groups. Subsequent Duncan tests were conducted to separate group means 17.
Results
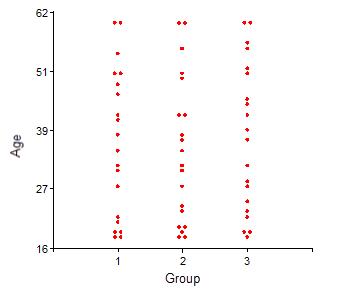
The sample included 50% males and 50% females. The ages ranged from 18 to 60 years. Fig. 1 shows the age distributions in each group under study, from which the sample equivalence can be determined. The Chi-square test yielded a value of χ2= 0.19 (p = 0.9958), indicating the homogeneous distribution of age categories between groups.
Mucin was detected in all the patients in the sample. Group 1 had a minimum salivary mucin value of 0.08 mg/ml, a maximum of 2.48 mg/ml, an average of 1.27 mg/ml, and a 0.63 mg/ml standard deviation. Group 2 had a minimum value of 1.20 mg/ml, a maximum value of 3.70 mg/ml, an average of 1.93 mg/ml, and a 0.61 mg/ml standard deviation. Group 3 had a minimum value of 1.40 mg/ml, a maximum value of 4.89 mg/ml, an average of 3.01 mg/ml, and a 1.03 mg/ml standard deviation. Fig. 2 shows the average mucin contents and their standard errors by study group.
The analysis of variance and subsequent F test (F = 25.01, p < 0.0001) confirm significant differences in mucin content. The results of Duncan’s test (Table 1) show that the mean mucin contents in Group 1 are lower than in the other two groups and that those of Group 2 are lower than those of Group 3.
Table 1: Average mucin contents by group studied and differences according to Duncan’s test
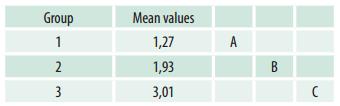
Mean values with a common letter are not significantly different (p > 0.05)
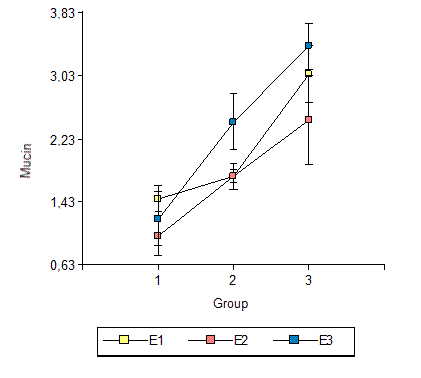
Fig. 3 shows the averages and standard errors of mucin contents by study group and gender. Although the increase in mucin content from Group 1 to Group 2 and from Group 2 to Group 3 appears to be more marked in males than in females, the F test (F = 3.1, p = 0.0856) did not detect a significant difference based on gender.
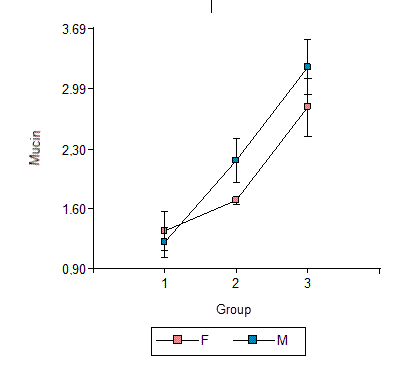
Fig. 3: Averages and standard errors of saliva mucin content in the three groups under study by gender.
The Pearson correlation coefficients when studying the association between mucin content and age were not significant in Groups 1 (r = -0.13, p = 0.5834) and 3 (r = 0.06, p = 0.8135), but were significant in Group 2 (r = 0.60, p = 0.005). This shows that patients with periodontitis have a higher saliva mucin content with increasing age. Fig. 4 shows the mean saliva mucin contents by study group and age.
Discussion
Despite advances in diagnostic tools, it is still not possible to predict how periodontal destruction will occur in each individual. Early clinical detection of the disease in patients with initial lesions is essential. This could be done through an indicator that is easy to access and read and that enables clinicians to detect affected areas or risk cases early so they can implement appropriate planning and/or prevention strategies.
As the disease involves mainly an inflammatory reaction, there should be chemical markers to detect possible attacks on the tissues. Therefore, some of the substances present in saliva, such as mucin, should be studied precisely at the local level.
This study determined salivary mucin activity as a biomarker in patients with periodontal disease. This study showed a marked increase in mucin concentration in the saliva of patients with gingivitis and even more in patients with periodontitis.
Normal mucin concentration in saliva is 1.19 mg/ml. Swati Kejriwal et al.15demonstrated an increase in salivary mucin concentration in patients with gingivitis and a decrease in mucin concentration in the periodontitis group compared to the gingivitis group. This is consistent with our study, as our results show a marked increase in salivary mucin concentration in patients with gingivitis. However, and unlike the findings of these authors, we found that patients with periodontitis had a higher salivary mucin concentration than patients with gingivitis.
Sánchez et al. 18 determined that protein, amylase, and mucin concentrations increased in subjects with moderate and severe periodontal disease in unstimulated saliva; this study yielded similar results regarding the increase in mucin concentration in patients with periodontitis.
Acquier et al. 19 found that salivary mucin concentration increases in patients with chronic periodontitis and aggressive periodontitis. This is consistent with our study in terms of increased salivary mucin concentration in patients with periodontitis.
Lundmark et al. 20 found a decrease in salivary mucin concentration in patients with periodontitis, contrary to what was found in this study, where mucin concentration in patients with periodontitis is the highest of the three groups studied.
Conclusions
Increased salivary mucin concentration in periodontal patients could represent a chemical marker useful to help clinically diagnose the disease.
There is a direct relationship between salivary mucin content and periodontal disease. Salivary mucin concentration is lower in patients who do not suffer from periodontal disease, increases in patients with gingivitis and increases even more in patients with periodontitis.
Gender did not affect salivary mucin content in the groups studied.
Salivary mucin content and patient age did not correlate significantly in patients with gingivitis or with no periodontal disease. Salivary mucin content in patients with periodontitis increases with age.
REFERENCES
1. Herrera D, Figuero E, Shapira L, Jin L, Sanz M. La Nueva clasificación de las enfermedades periodontales y periimplantarias. Rev Cient Soc Esp Period. 2018;1(9):94-110. [ Links ]
2. Martínez LL, López MDCGC, Rodríguez AWH, Sierra EMS, Luna MAL, Rodríguez SHS. La caries, gingivitis, periodontitis y la maloclusión siguen siendo las afecciones estomatológicas más frecuentes en la población. Arch Med. 2014;9(4). [ Links ]
3. Lindhe J, Karring T, Lang NP. Periodontología clínica e implantología odontológica. 5 ed. Educ Med Panamericana; 2009 289 p. [ Links ]
4. Newman MG, Takei H, Klokkevold PR, Carranza FA. Carranza's clinical periodontology. 11 ed. Elsevier health sciences; 2011 176 p. [ Links ]
5. Romero C, Suarez M, Gloria-Narváez C. Proteínas Totales, Fosfatasa Alcalina, Prostaglandinas E2 y Lisozima como Biomarcadores Salivales en Pacientes Adultos con Periodontitis Crónica. Int J Odontostomatol. 2017;11(4):381-5. [ Links ]
6. Villacís Tapia ÁF 2017. Niveles de la enzima alanino aminotransferasa en saliva en periodontitis crónica y su relación con la severidad (Master's thesis, Quito: UCE). [ Links ]
7. Baudo JE, Tosti SB, Mazzeo DMA, Cecho AC, Allegretti PE. IL-1 y TNF a como biomarcadores salivares de enfermedad periodontal. Rev Fac Odontol. 2015;2015. [ Links ]
8. Johansson MEV, Hansson GC. The mucins. Encyclopedia of immunobiology. 2016. p381-8 [ Links ]
9. Hernández Castañeda AA, Aranzazu Moya GC. Características y propiedades físico-químicas de la saliva: una revisión. UstaSalud. 2012;11(2):102-12. doi: 10.15332/us.v11i2.1123. [ Links ]
10. Gésime JM, Acevedo AM, Lalaguna F. Las mucinas salivales y sus implicaciones en la reología de la saliva humana y los sustitutos salivales. Acta Odontol Venez. 2009;47(2):446-52. [ Links ]
11. Dhanisha SS, Guruvayoorappan C, Drishya S, Abeesh P. Mucins: structural diversity, biosynthesis, its role in pathogenesis and as possible therapeutic targets. Crit Rev Oncol Hematol. 2018; 122:98-122. doi: 10.1016/j.critrevonc.2017.12.006 [ Links ]
12. Diajil AR, Sood LI, Azeez RA. A Salivary a-amylase Level in Relation to the Oral Health Parameters among Children in Baghdad City. Journal of baghdad college of dentistry. 2016;28(2):40-6. [ Links ]
13. Daza ANZ, Huerta ERL, Martínez FF. Determinación de pH y proteínas totales en saliva en pacientes con y sin aparatología ortodóncica fija (estudio piloto). Rev Odontol Mex. 2004;8(3):59-63. [ Links ]
14. Slomiany BL, Murty VLN, Piotrowski J, Slomiany A. Salivary mucins in oral mucosal defense. Gen Pharmacol Vasc Syst. 1996;27(5):761-71. doi: 10.1016/0306-3623(95)02050-0 [ Links ]
15. Sarosiek J, Rourk RM, Piascik R, Namiot Z, Hetzel DP, McCallum RW. The effect of esophageal mechanical and chemical stimuli on salivary mucin secretion in healthy individuals. Am J Med Sci. 1994;308(1):23-31. doi: 10.1097/00000441-199407000-00006 [ Links ]
16. Kejriwal S, Bhandary R, Thomas B, Kumari S. Estimation of levels of salivary mucin, amylase and total protein in gingivitis and chronic periodontitis patients. J Clin Diagn Res. 2014;8(10): ZC56-60. doi: 10.7860/JCDR/2014/8239.5042 [ Links ]
17. Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW. InfoStat versión 2020. Argentina: Grupo InfoStat, FCA, Universidad Nacional de Córdoba; 2020. Disponible en: http://www.infostat.com.ar. [ Links ]
18. Perelman SB, Garibaldi LA, Tognetti PM. Experimentación y modelos Estadísticos. Buenos Aires: Facultad de Agronomía. Universidad de Buenos Aires; 2019. 475 p. [ Links ]
19. Sánchez GA, Miozza V, Delgado A, Busch L. Determinación de niveles salivales de mucina y amilasa en pacientes con periodontitis crónica. J Periodont Res. 2011;46(2):221-7. doi: 10.1111/j.1600-0765.2010.01332.x. [ Links ]
20. Acquier AB, PITA AKDC, Busch L, SÁNCHEZ GA. Comparison of salivary levels of mucin and amylase and their relation with clinical parameters obtained from patients with aggressive and chronic periodontal disease. J Appl Oral Sci. 2015;23(3):288-94. doi: 10.1590/1678-775720140458 [ Links ]
Authorship contribution 1. Conception and design of study 2. Acquisition of data 3. Data analysis 4. Discussion of results 5. Drafting of the manuscript 6. Approval of the final version of the manuscript. MJA has contributed in: a,b,c,d,e,f. RPJ has contributed in: a,d,e,f.
Received: March 04, 2021; Accepted: April 12, 2021










 texto em
texto em