Serviços Personalizados
Journal
Artigo
Links relacionados
Compartilhar
Odontoestomatología
versão impressa ISSN 0797-0374versão On-line ISSN 1688-9339
Odontoestomatología vol.22 no.36 Montevideo 2020 Epub 01-Dez-2020
https://doi.org/10.22592/ode2020n36a5
Research
Profile of oral squamous cell carcinoma at the Anatomic Pathology Laboratory of the School of Dentistry of Universidad de la República, 1982-2015 period
1Cátedra de Anatomía Patológica. Facultad de Odontología, Universidad de la República, Montevideo, Uruguay
2Cátedra de Anatomía Patológica. Facultad de Odontología , Universidad de la República, Montevideo, Uruguay
Oral squamous cell carcinoma is the most frequent malignant oral pathology, mainly associated with tobacco, alcohol, and human papillomavirus.
This study aimed to determine the profile of this pathology at the Laboratory of Anatomic Pathology of the School of Dentistry of Universidad de la República between 1982 and 2015. A retrospective, cross-sectional, analytical study was conducted. Statistical analysis performed using the Cox model, Fisher’s exact test, and the Kaplan-Meier method. Of 207 cases, 59.9% were men and 98.5% older than 40. It is usually located on the tongue, with a predominance of histopathologically well and moderately differentiated carcinomas. There was an association between histopathological grade, gender, and age, with an average survival time of 2.36 years and a 43% five-year survival rate. The study allowed us to determine the situation of this pathology in a reference health service and create a database for subsequent studies.
Keywords: head and neck squamous cell carcinoma; epidemiology; survival
El Carcinoma oral de células escamosas es la patología oral maligna más frecuente, asociada principalmente al tabaco, alcohol y virus del papiloma humano.
El objetivo del estudio fue determinar el perfil de dicha patología, en el Laboratorio de Anatomía Patológica de la Facultad de Odontología de la Universidad de la República entre 1982-2015. Se realizó un estudio retrospectivo, transversal, analítico. Análisis estadístico usando el modelo de Cox, el test exacto de Fisher y el método de Kaplan Meier. Con 207 casos el 59,9% fueron hombres y el 98.5% mayores de 40 años. Localizándose preferentemente en la lengua, con predominio de los carcinomas histopatologicamente bien y moderadamente diferenciados. Se encontró asociación entre el grado histopatológico, género y edad; con un tiempo medio de sobrevida de 2.36 años y una tasa de sobrevida del 43% a los 5 años. El estudio permitió conocer dicha patología en un servicio de referencia y generar una base de datos para posteriores estudios.
Palabras claves: Carcinoma de células escamosas de cabeza y cuello; Epidemiología y Sobrevida
O carcinoma epidermóide de boca é a patologia maligna mais frequente, principalmente associada ao tabaco, álcool e papiloma vírus humano.
O objetivo do estudo foi determinar o perfil dessa patologia, no Laboratório de Anatomia Patológica da Faculdade de Odontologia da Universidade da República entre 1982-2015. Foi realizado um estudo analítico retrospectivo, transversal. Análise estatística pelo modelo de Cox, teste exato de Fisher e método de Kaplan Meier. Em 207 casos, 59,9% eram homens e 98,5% tinham mais de 40 anos. De preferência localizado na língua, com predominância de carcinomas histopatologicamente bem e moderadamente diferenciados. Foi encontrada associação entre grau histopatológico, sexo e idade; com tempo médio de sobrevivência de 2,36 anos e taxa de sobrevivência de 43% aos 5 anos. O estudo permitiu conhecer essa patologia em um serviço de referência e gerar um banco de dados para estudos subsequentes.
Palavras-chave: Carcinoma espinocelular de cabeça e pescoço; Epidemiologia e Sobrevivência
Introduction and background
Oral squamous cell carcinoma (OSCC) is the most frequent oral malignancy, accounting for 2%-3% of all malignancies and up to 80%-90% of those of the oral cavity 1-8.
OSCC is a solid malignant tumor originating in the stratified squamous epithelium of the upper aerodigestive tract’s mucosa, characterized by varying degrees of histological differentiation and a high invasive and metastatic potential9. Its etiology is mainly associated with risk factors such as tobacco, alcohol, and human papillomavirus 16 and 18 (HPV) 10-13.
In 2018, the Global Cancer Observatory (GLOBOCAN) reported the following data for Uruguay: 15,101 new cancer cases in a population of 3,469,551 people, with 8,589 deaths and a 5-year prevalence of 35,478 cases, according to sources from the National Cancer Registry (NCR) and the World Health Organization (WHO) 14.
The cancer epidemiological situation of Uruguay-recently published by the NCR- reported the following data on oropharyngeal cancer for 2011-2015 (expressed in adjusted rates per 100,000 inhabitants): a total of 1,529 new cases (1,107 men and 422 women), mainly on the tongue (378 cases) regardless of lingual anatomical site, an incidence of 10.17 and 2.81 for men and women respectively, and a 6.67 mortality rate in men and of 1.21 in women15.
Morphologically, OSCC is a malignant epithelial neoplastic proliferation, consisting of nests, cords, and islets that resemble-to a greater or lesser degree-the original squamous epithelium of the lining mucosa. Currently, OSCC histological grading is based on the WHO’s histological classification and the tumor-invasive front: well differentiated, moderately differentiated, and undifferentiated OSCC 7-8.
OSCC is an aggressive neoplasm with unpredictable biological behavior and an unfavorable prognosis16. The aggressiveness of this malignancy is evident, as despite currently available therapeutic strategies-including surgical removal either exclusively or in combination with radiation therapy and/or chemotherapy -the survival and recurrence rates after five years is poor, where only 50% of the patients diagnosed and treated survive17-19, a situation that has remained unchanged over the last five decades 20-22. Its prognosis is related to factors such as tumor size, presence of metastatic lymph nodes, tumor differentiation, invasion of adjacent structures, location, and treatment 23-24. The TNM staging system, where T stands for primary tumor, N for regional lymph nodes and M for distant metastases, is a universal method of staging the disease in cancer patients, which allows clinicians to make therapeutic decisions. It was updated in 2017 for each topography, in addition to identifying OSCC HPV+ and HPV 25.
This study aimed to determine this pathology profile at the Laboratory of Anatomic Pathology of the School of Dentistry (LAPFO) of Universidad de la República between 1982 and 2015. It also aimed to describe its distribution according to clinical-morphological parameters (age, gender, location, and histopathological grade) and determine its survival rate.
Methodology
1- Ethical considerations
This study was conducted with the approval of the Ethics Committee of the School of Dentistry of Universidad de la República, Montevideo, Uruguay (File 163/16).
2- Study design
A retrospective, cross-sectional and analytical study, with convenience non-probability sampling according to the proposed inclusion and exclusion criteria. The population included all of LAPFO’s biopsy records (11,321) for 1982-2015. The sample included all the cases with histopathological diagnosis of OSCC of the oral mucosa. A sample of 207 OSCC cases (1.8%) was obtained from the total biopsy records for that period (11,321); they met the inclusion and exclusion criteria set out below.
3- Inclusion and exclusion criteria
Inclusion criteria: OSCC with full morphological, clinical data, and recorded variables: age, gender, location, and histopathological grade of the lesion. The OSCC histopathological diagnosis accepted was that recorded in the biopsy report that met the WHO’s histological criteria of malignancy (well differentiated, moderately differentiated, and undifferentiated). Reassessing the histopathological diagnosis was considered when the grade was not detailed according to the established criteria, but it was not necessary in any case. If there were two records (incisional and excisional biopsy) for one patient, it was considered a single case-to avoid duplication-and the histological grading recorded was the one for the excisional biopsy. In contrast, for the date of onset of the disease, the one recorded in the first biopsy with an OSCC diagnosis was considered.
A sample was used to determine the survival rate of OSCC patients; the inclusion criterion was that the record should include the patient’s identity card number, which was necessary to find the patient in the NCR’s database (n=99).
Exclusion criteria: Cases with incomplete data, missing variables (age, gender, location, and histological grading) were excluded.
4- Statistical analysis
Descriptive statistics were used to establish significant relationships between the different clinical-morphological parameters through Fisher’s exact test. Survival curves were created using Kaplan Meier’s methodology. Hypothesis tests were performed at a 5% level, and Survival analysis using software R. The data were analyzed using the Cox proportional-hazards model and based on the potential influence of gender, age, histopathological grade, and clustered location of tumors. The variables were selected with a backward procedure; after adjusting the models, the validity of the proportionality assumption was verified.
Results
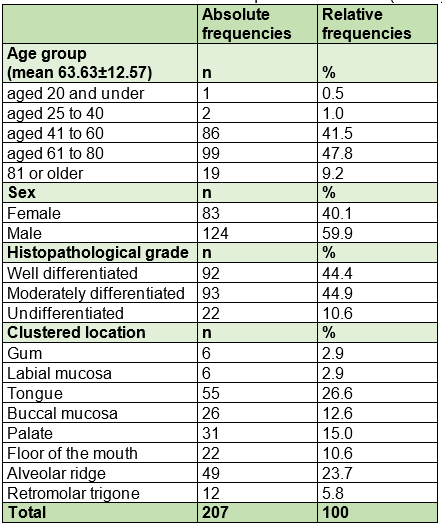
The results are shown in Chart 1. An age range between 16 and 99 was recorded, with an average of 63 years ± 12.57, 41.5% (86) between 41 and 60, and 47.8% (99) between 61 and 80. In the sample, 59.9% (124) were male, with a male: female ratio of 1.5:1. Regarding histopathological grade, 44.4% (92) were well differentiated carcinomas, 44.9% (93) moderately differentiated and 10.6% (22) undifferentiated. The most frequent location was the tongue-26.6% (55)-and the alveolar ridge-23.7% (49).
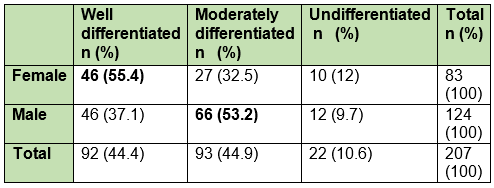
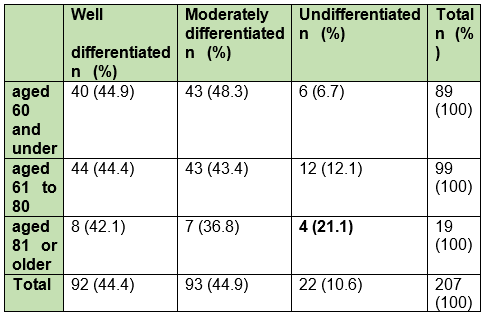
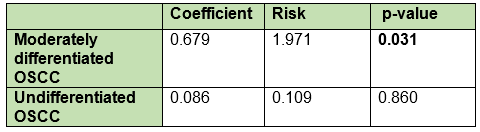
Variable correlation showed that the histopathological grade concerning gender and age was significant, showing that men have a higher rate of moderately differentiated OSCC and women, a higher rate of well-differentiated OSCC (p-value=0.031) (Table 1); as for age, those over 81 have a higher rate of undifferentiated OSCC (p-value=0.027) (Table 2).
Multivariate analysis of location showed no significant association, possibly due to the multiple locations of OSCC in the oral mucosa. In this study, locations were grouped into eight categories. In women, the most frequent locations were the alveolar ridge-28.9% (24)-and the tongue-26.5% (22)-while in men, OSCC appeared in the tongue in 26.6% of cases (33), and in 20.2% of cases (25), in the alveolar ridge.
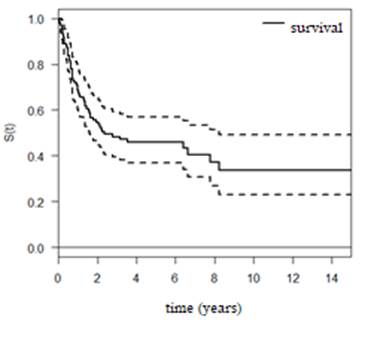
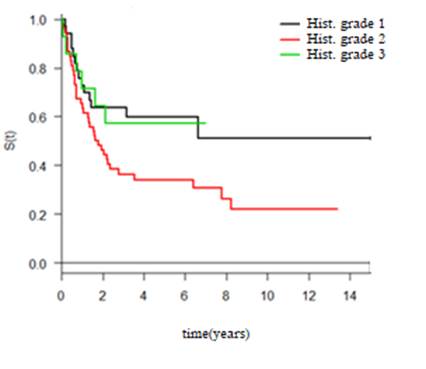
Survival analysis was performed with a sample of 99 patients, among whom death occurred in 57 cases; the rest corresponded to censoring. The survival curve (Figure 1) was created, from which it was estimated that the average survival time was 2.36 years with a confidence interval (1.60-8.24), and the five-year survival rate survival was 43%, without considering the stages of the TNM system as they were not included in the records.
According to gender, age, histopathological grade, and location, a statistically significant correlation was found with the histological grade when associating the risk of death. The final model is presented in Chart 2, which shows that patients diagnosed with moderately differentiated OSCC have almost twice the risk of death than patients with well differentiated OSCC (p-value 0.031). When analyzing undifferentiated OSCCs, death occurred in all cases, approximately seven years after follow-up, unlike well differentiated OSCCs, which at the end of the study still had patients that were alive (Fig. 2).
Discussion
It is essential to study OSCC due to its high morbidity and mortality. It is instrumental to obtain relevant information to compare results with other centers specialized in oral pathology, providing knowledge on survival and clinical-pathological characteristics.
This study made it possible to obtain institutional information for the School of Dentistry of Universidad de la República, establishing the OSCC profile at LAPFO; this allowed us to create a significant database for future studies.
This study had some limitations given the lack of proper anatomical-pathological recording, making TNM staging, and risk factor assessment difficult. Still, it was significant to have a cohort of 99 patients for survival analysis.
In this study, 59.9% of OSCC cases affected men, a figure in line with the literature 19,21,26-27; however, differences were found between the m:f ratio of our study (1.5:1) and that reported by the National Cancer Institute (INCA) in Brazil, which reported a rate higher than 3.7:1 28 and that recently reported by the NCR in Uruguay, which published a 3.6:1 ratio for both men and women 15. The highest frequency among men matches the data reported in GLOBOCAN 2018 14 and by other authors, where OSCC is two or three times more frequent in men in most ethnic groups. In recent years, there has been an increasing trend of OSCC among women due to increased tobacco consumption. This data could explain the 1.5:1 ratio of our study 2,19,29.
A retrospective study published in 2014 by Momares et al., including 217 patients diagnosed with OSCC between 1989 and 2010 and conducted at the Department of Oral Pathology of the School of Dentistry of Universidad Mayor de Chile, showed similar results to those of our study. The results were compared, and it was found that distribution by gender, ratio between genders, age range, and average age were almost identical. The tongue and alveolar ridge were the most frequent locations for both, with some differences in the remaining locations. Regarding histological differentiation, both studies found that well differentiated and moderately differentiated OSCCs were more prevalent than undifferentiated OSCCs 30.
Regarding the age of appearance of OSCC in our study, the mean age was 63±12.57, with an age range of 16 to 99, an OSCC frequency of 41.5% between 41 and 60, increasing to 47.8% in the 61-80 age group. Oliveira et al. in 2015 reported an average age of 60.75±11.26 years in a public hospital in Uruguay, similar to the one observed in this study(19). The literature reports that over 90% of cases are individuals over 40, and more than 50% occur in individuals over 65. In our study, 98.5% occurred in individuals over 40, and 57% in individuals over 60 31-33.
Regarding the onset age of OSCC according to gender, 80% of women and 96% of men are aged between 41 and 80; of those groups, over 50% are in the highest range (61 to 80). In the age range analysis, we found no OSCC records for men in the group under 40, while in the group over 81, women recorded a figure almost three times higher than men.
The most frequent location was the tongue-26.6%-in line with a study conducted at the Laboratory of Anatomic Pathology of INCA of Uruguay in 2017, as reported in the literature in most OSCC studies 34. There is a difference regarding the second most frequent location (alveolar ridge: 23.7%), which most studies report to be the floor of the mouth and/or palate. This may be linked to how OSCC locations are grouped 35-37.
Our histopathological grading results are similar to those obtained by Wei-ping Jie et al.: 90.8% of the cases reported were well differentiated and moderately differentiated OSCC 38. Other studies showed similar data. Sairay et al. showed that 52% of OSCC were moderately differentiated and 42% well differentiated in Pakistan and for the 2015-2016 period 39, while Gul et al. found that 43% of patients in a tertiary hospital in southern Punjab India had well differentiated OSCC, 38%, moderately differentiated OSCC, and 20%, undifferentiated OSCC 40. In 2018, Read et al. studied OSCC in a Brazilian population with low economic development, with n=194 cases; 54.6% were well differentiated, 37.1%, moderately differentiated, and 5.2%, undifferentiated carcinomas. Therefore, this study has a similar profile to that of our study 41.
When connecting variables, we found a statistical significance in OSCC histopathological grade in relation to gender and age. Men had a higher rate of moderately differentiated carcinomas, and women a higher rate of well differentiated OSCC. This indicates that the rate of undifferentiated carcinomas increases with age. This matches the results of a study published in 2010 on the clinical-histopathological characteristics of OSCC at San Vicente de Paúl University Hospital, Colombia 42.
The five-year survival rate of OSCC patients was 43%, similar to that reported by Momares et al. in Chile, which was 46%, while their average survival time was 6.9 years, higher than that found in our study, which was 2.36 years 30. Other authors report a survival rate ranging from 30% to 50% for OSCC33-43.
Oliveira et al. show an even lower overall survival rate, with a 24% five-year survival 19. In 2014, Bonfante et al. conducted a national study of Brazil’s Unified Health System for 2000-2006. They reported a five-year survival rate of 4.6-years for mouth cancer, and a five-year overall survival rate of 2.6-years, similar to our findings (2.36 years) 44. Another study with 274 cases from the University of Buenos Aires (Argentina) evaluated overall survival for oral cancer and reported a five-year survival rate of 39% 45.
Conclusions
In general, OSCC studies in Uruguay are scarce, mainly regarding patient prognosis and survival. Retrospective studies such as this one have significant limitations given the lack of complete records, missing-in most cases-relevant clinical data to establish a TNM staging system, and mainly due to the lack of patient follow-up. However, these descriptive studies are a source of hypotheses for further analytical studies.
Considering the high mortality and morbidity of OSCC, health professionals should consider the importance of early detection of these injuries and education on known risk factors, such as tobacco, alcohol, and HPV infection, bearing in mind that late diagnosis is one of the main factors concerning poor prognosis.
REFERENCES
1. Maraboli-Contreras S, Adorno-Farias D, Maturana-Ramirez A, Rojas-Alcayaga G, Fuentes-Alburquenque M, Espinoza-SantanderI. Sobrevida de carcinoma oral de células escamosas: reporte de la Universidad de Chile.Rev. Clin. Periodoncia Implantol. Rehabil. Oral. 2018; 11 (3): 147-151. [ Links ]
2. Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015; 65 (2): 87-108. [ Links ]
3. Warnakulasuriya S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncology. 2009; 45: 309-316. [ Links ]
4. Neville BW. Patologia oral e maxilofacial. 4. ed. Rio de Janeiro: Elsevier, 2016. 912 p. [ Links ]
5. Licitra L, Locati LD, Bossi P, Cantù G. Head and neck tumors other than squamous cell carcinoma. Curr Opin Oncol. 2004; 16 (3): 236-41. [ Links ]
6. Johnson NW, Jayasekara P, Amarasinghe AAHK. Squamous cell carcinoma and precursor lesions of the oral cavity: epidemiology and etiology. Periodontol 2000. 2011 ; 57 (1): 19-37. [ Links ]
7. Barnes L, Eveson JW, Reichart P, Sidransky D. World Health Organization classification of tumours: Pathology and genetics Head and Neck tumours. International Agency for Research on Cancer, Lyon, France 2005; 168-175 [ Links ]
8. El-Naggar AK, Chan JKC, Rubin Grandis J, Takata T, Slootweg PJ. WHO classification of head and neck tumours. International Agency for Research on Cancer. 4th ed. 2017. [ Links ]
9. Kang H, Kiess A, Chung C. Emerging biomarkers in head and neck cancer in the era of genomics. Nat Rev Clin Oncol. 2015; 12: 11-26. [ Links ]
10. Miguel Cruz PA, Niño Peña A, Batista Marrero K, Miguel-Soca PE. Factores de riesgo de cáncer bucal. Rev Cubana Estomatol. 2016; 53 (3): 128-45. [ Links ]
11. Jiang X, Wu J, Jiexue Wang RH. Tobacco and oral squamous cell carcinoma: A review of carcinogenic pathways. Tob Induc Dis. 2019; 17. [ Links ]
12. Auluck A, Hislop G, Bajdik C, Poh C, Zhang L RM. Trends in oro- pharyngeal and oral cavity cancer incidence of human papillomavirus (HPV)-related and HPV-unrelated sites in a multicultural population: the British Columbia experience. Cancer. 2010; 116: 2635-2644. [ Links ]
13. Young D, Xiao CC, Murphy B, Moore M, Fakhry C, Day TA. Increase in head and neck cancer in younger patients due to human papillomavirus (HPV). Oral Oncology. 2015; 51. [ Links ]
14. Ferlay J, Colombet M, Soerjomataram I, Mathers C, Parkin DM, Piñeros M, Znaor A, Bray F. Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2018. https://doi.org/10.1002/ijc.31937 [ Links ]
15. Comisión Honoraria de Lucha Contra el Cáncer. Situacion Epidemiologica del Uruguay en relacion al Cancer. Regist Nac del Cáncer, Com Honor Lucha Contra el Cáncer. 2019;1-61. [ Links ]
16. Lopes KM,Souza JA, Lacerda de Souza L, Akiko ML, Guimarão DM, Pontes HA . La proteína Ki-67 predice la supervivencia en las células de carcinoma escamoso oral: un estudio inmunohistoquímico. Braz. res oral. 2017; 31: e66. [ Links ]
17. Majumdar B, Shankargouda P, Sachin CS, Gargi SS and Roopa SR. Clinico-pathological prognosticators in oral squamous cell carcinoma: An update Translational. Research in Oral Oncology. 2017; (2): 1-14. [ Links ]
18. Mamelle G, Pampurik J, Luboinski B, Lancar R, Lusinchi A, Bosq J. Lymph node prognostic factors in head and neck squamous cell carcinomas. Am J Surg. 1994; 168 (5): 494-8. [ Links ]
19. Oliveira MLC, Wagner VP, Sant'ana Filho M, Carrard VC, Hugo FN, Martins MD. A 10-year analysis of the oral squamous cell carcinoma profile in patients from public health centers in Uruguay. Braz Oral Res. 2015; 29 (1): 1-8. [ Links ]
20. Barrios E, Garau M. Cáncer: magnitud del problema en el mundo y en Uruguay, aspectos epidemiológicos. An la Fac Med. 2017; 4 (1): 04-66. [ Links ]
21. Barrios E, Garau M, Alonso R, Musetti C. IV atlas de incidencia del cancer en el Uruguay. Registro Nacional de Cáncer, Comisión Honoraria de lucha contra el Cáncer, 2014. [ Links ]
22. Ferlay J, Soerjomataram I, Dikshit R, Eser S, Mathers C, Rebelo M, et al. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015; 136 (5). [ Links ]
23. Patel RS, Clark JR, Dirven R, Wyten R, Gao K, O'Brien CJ. Prognostic factors in the surgical treatment of patients with oral carcinoma. ANZ J Surg. 2009 Jan;79(1-2):19-22. [ Links ]
24. Mamelle G, Pampurik J, Luboinski B, Lancar R, Lusinchi A, Bosq J. Lymph node prognostic factors in head and neck squamous cell carcinomas. Am J Surg. 1994;168(5):494-8. [ Links ]
25. Lydiatt WM, Patel SG, O'Sullivan B, Brandwein MS, Ridge JA, Migliacci JC, et al. Head and neck cancers-major changes in the American Joint Committee on cancer eighth edition cancer staging manual. CA Cancer J Clin. 2017; 67(2): 122-37. [ Links ]
26. Roberto De Oliveira L, Ribeiro-Silva A, Zucoloto S. Perfil da incidência e da sobrevida de pacientes com carcinoma epidermóide oral em uma população brasileira Incidence and survival profile of patients with oral squamous cell carcinoma in a Brazilian population. J Bras Patol Med Lab. 2006; 42 (5): 385-92. [ Links ]
27. Ferlay J, Colombet M, Soerjomataram I, Mathers C, Parkin DM, Piñeros M, Znaor A ,Bray F.Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer. 2018; 144 (8): 1941-1953 [ Links ]
28. Estimativa 2018: incidência de câncer no Brasil / Instituto Nacional de Câncer José Alencar Gomes da Silva. In 2017. Ministério da saúde, Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). [ Links ]
29. Chan MH, Wang F, Mang W kong, Tse LA. Sex Differences in Time Trends on Incidence Rates of Oropharyngeal and Oral Cavity Cancers in Hong Kong. Ann Otol Rhinol Laryngol. 2018; 127 (12): 895-902. [ Links ]
30. Momares B, Contreras G, Martínez B, Ávalos N, Carmona L. Sobrevida en Carcinoma Espinocelular de mucosa oral: análisis de 161 pacientes. Rev Chil Cir. 2014; 66 (6): 568-76. [ Links ]
31. Shridhar K, Rajaraman P, Koyande S, Parikh PM, Chaturvedi P, Dhillon PK, Rajesh Dikshit P. Trends in mouth cancer incidence in Mumbai, India (1995-2009): An age-period-cohort analysis. Trends mouth cancer Incid Mumbai. 2016; 42 (2016): 66-71. [ Links ]
32. Salgado-Ramíre B, Rivera-Martínez CG, Altamirano-Díaz I, Salgado-Ramírez B. Frecuencia de carcinoma de cavidad oral en un Hospital de tercer nivel. Rev Sanid Milit Mex. 2014; 68 (3): 172-6. [ Links ]
33. Álvarez Arias D, Munyo Estefan A, Borche G, Rodríguez Sande V, Cuello M. Cáncer de cabeza y cuello en Uruguay. Análisis de sobrevida en dos centros de referencia. Rev Méd Urug. 2018; 34 (1): 21-8. [ Links ]
34. Beovide Cortegoso AV, Koerich Laureano N, Dutra da Silva A, Krebs Danilevicz C, Sellinger Magnusson A, et al. Cell proliferation markers at the invasive tumor front of oral squamous cell carcinoma: comparative analysis in relation to clinicopathological parameters of patients. J Appl Oral Sci. 2017; 25 (3): 318-23. [ Links ]
35. Brandizzi D, Gandolfo M, Velazco ML, Cabrini RL, Lanfranchi HE. Clinical features and evolution of oral cancer: A study of 274 cases in Buenos Aires, Argentina. Med Oral Patol Oral Cir Bucal. 2008; 13 (9): E544-8. [ Links ]
36. Siriwardena BS, Tilakaratne A, Amaratunga EAPD, Tilakaratne WM. Demographic, aetiological and survival differences of oral squamous cell carcinoma in the young and the old in Sri Lanka. Oral Oncol. 2006; 42 (8): 831-6. [ Links ]
37. Muradás Girardi F, Fuga GR, Callai T, Fernandes Bianchi M, Girardi FM. Histopatological differences between genders in patients witH oral squamous cell carcinoma. Clin Biomed Res. 2017;37 (3): 147-50. [ Links ]
38. Jie W, Bai J, Li B. Clinicopathologic Analysis of Oral Squamous Cell Carcinoma After 125 I Interstitial Brachytherapy. Technol Cancer Res Treatmen. 2018;17:1-5. [ Links ]
39. Saira Fiaz Khan M, Rauf Khattak M, Rawail A, Malik S. Epidemiological and clinical correlates of oral squamous cell carcinoma in patients from north-west Pakistan. J Pak Med Assoc. 2019; 69 (8): 1074-1078 [ Links ]
40. Gul H, Asif F, Ghaffar I, Anwar MA, Tayyab MA, Kashif M. Epidemiology and pathological trends in oral squamous cell carcinoma in a local tertiary care hospital. Int J Community Med Public Heal. 2017; 4 (12): 4440. [ Links ]
41. Leite AA, Leonel ACL da S, de Castro JFL, Carvalho EJ de A, Vargas PA, Kowalski LP, da Cruz Perez DE. Oral squamous cell carcinoma: A clinicopathological study on 194 cases in northeastern Brazil. A cross-sectional retrospective study. Sao Paulo Med J. 2018; 136 (2):165-9. [ Links ]
42. Álvarez Martínez E, Preciado A, Montoya Fernández SA, Jiménez Gómez R, Posada A. Características clínico-histopatológicas del carcinoma escamocelular bucal, Colombia. Rev Cubana Estomatol. 2010; 47 (1): 81-95. [ Links ]
43. Bórquez PM, Capdeville FF, Madrid AM, Veloso MO, Cárcamo MP. Sobrevida global y por estadíos de 137 pacientes con cáncer intraoral. Experiencia del Instituto Nacional del Cáncer.Rev. Chilena de Cirugía.2011; 63 (4): 351-355. [ Links ]
44. Bonfante GM, Machado CJ, Souza PEA, Andrade EIG, Acurcio F de A, Cherchiglia ML. Sobrevida de cinco anos e fatores associados ao câncer de boca para pacientes em tratamento oncológico ambulatorial pelo Sistema Único de Saúde, Brasil. Cad Saude Publica. 2014; 30 (5): 983-97. [ Links ]
45. Brandizzi D, Gandolfo M, Velazco ML CR, Lanfranchi HE. Clinical features and evolution of oral cancer: a study of 274 cases in Buenos Aires, Argentina.Med Oral Patol Oral Cir Bucal. 2008;13:E544-8. [ Links ]
Authorship contribution 1) Conception and design of study 2) Acquisition of data 3) Data analysis 4) Discussion of results 5) Drafting of the manuscript 6) Approval of the final version of the manuscript NGU has contributed in 1, 2, 3, 4, 5, and 6. VBC has contributed in 1, 4, 5, and 6.
Received: October 29, 2019; Accepted: September 08, 2020










 texto em
texto em