Serviços Personalizados
Journal
Artigo
Links relacionados
Compartilhar
Odontoestomatología
versão impressa ISSN 0797-0374versão On-line ISSN 1688-9339
Odontoestomatología vol.21 no.34 Montevideo dez. 2019 Epub 01-Dez-2019
https://doi.org/10.22592/ode2019n34a2
Research
Effect of EDTAC and citric acid on dentinal walls. Variation of exposure time and dental age
1 Cátedra de Endodoncia, Facultad de Odontología, Universidad de la República, Uruguay. sylmarbaum@gmail.com
2 Cátedra de Endodoncia, Facultad de Odontología, Universidad de la República, Uruguay.
3 Ex docente de la Cátedra de Endodoncia, Facultad de Odontología, Universidad de la República, Uruguay.
The aim of this study was to find a formula and application time for chelators effective in cleaning the root canal without causing erosion and considering dental age. The sample included 120 teeth: 60 taken from young patients and 60 from adult patients. They were instrumented and irrigated with 2.5% sodium hypochlorite (NaOCl). Each subgroup was divided randomly into six groups: 4 groups of 12 teeth and 2 control groups of 6 teeth. In the final irrigation, 17% ethylenediaminetetraacetic acid plus Cetavlon (EDTAC) or 10% citric acid was applied for 1 or 3 minutes according to each group. The results showed a high level of cleanliness, and a few showed erosion of the dentinal tubules. Satisfactory results were obtained in the removal of the smear layer when applying EDTAC or citric acid combined with NaOCl. Erosion was present in different degrees regardless of dental age, but it was indeed affected by the irrigant exposure time.
Keywords: chelators; smear layer; dentin permeability; erosion
El objetivo de este estudio fue lograr una fórmula y un tiempo de aplicación de los quelantes efectivos que logren la limpieza de las paredes del conducto sin causar erosiones, teniendo en cuenta la edad dentaria. La muestra constó de 120 dientes, 60 de pacientes jóvenes y 60 de pacientes adultos, se instrumentaron e irrigaron con hipoclorito de sodio (NaOCl) 2,5%. Cada subgrupo fue dividido al azar en 6 grupos: 4 grupos de 12 dientes y 2 grupos control con 6 dientes cada uno. En la irrigación final se aplicó ácido etilendiaminotetraacético más cetavlón (EDTAC) 17% o ácido cítrico 10% durante 1 o 3 minutos según el grupo. Los resultados presentaron buen nivel de limpieza con algunas muestras con erosión de los túbulos dentinarios. Con EDTAC o ácido cítrico combinado con NaOCl se lograron resultados satisfactorios en la remoción del barro dentinario. La erosión estuvo presente en sus diferentes grados independientemente de la edad dentaria, pero no del tiempo de exposición del irrigante.
Palabras clave: Quelantes; Barro dentinario; Permeabilidad de la dentina; Erosión
O objetivo deste estudo foi obter uma fórmula e um tempo de aplicação dos quelantes efetivos que permitam a limpeza das paredes do canal sem causar erosões, levando em consideração a idade dental. A amostra foi composta por 120 dentes, 60 de pacientes jovens e 60 de pacientes adultos, foram instrumentados e irrigados com hipoclorito de sódio (NaOCl) a 2,5%. Cada subgrupo foi dividido aleatoriamente em 6 grupos: 4 grupos de 12 dentes e 2 grupos de controle com 6 dentes cada um. Na irrigação final, ácido etilenodiaminotetracético com cetavlon (EDTAC) 17% ou ácido cítrico 10% foi aplicado por 1 ou 3 minutos, dependendo do grupo. Os resultados mostraram um bom nível de limpeza com algumas amostras com erosão dos túbulos dentinários. Com EDTAC ou ácido cítrico combinado com NaOCl, resultados satisfatórios foram obtidos na remoção da camada de esfregaço . A erosão esteve presente em seus diferentes graus independentemente da idade dentária, mas não do tempo de exposição do irrigante.
Palavras-chave: Quelantes; Camada de esfregaço; Permeabilidade da dentina; Erosão.
Introduction
During instrumentation of the root canal, an irregular and granular smear layer remains, resulting from filing the dental walls and organic remnants present in the canal 1; this layer is not observed on uninstrumented surfaces 2. This layer obstructs the dentinal tubules at a depth of 30 to 40 µm 3 by decreasing dentinal permeability, causing a delay in the action of topical medication and irrigants 4-5 and preventing close contact of the filling material with the dentinal walls 1,3-4. Consequently, there is a higher risk of bacterial infection and microfiltration 3,6).
The conventional irrigation protocol includes sodium hypochlorite (NaOCl) at 2.5% to 5.25% concentrations and a chelator; they act on the organic and inorganic part of the smear layer, but also on the canal wall, causing its demineralization 2,5,7.
The most commonly used chelators are ethylenediaminetetraacetic acid (EDTA) and citric acid. In 1957, Ostby8 introduced EDTA disodium salt as an auxiliary irrigant in narrow and/or calcified canals. Its original formula consisted of 17 g EDTA, 05N sodium hydroxide (NaOH) and 100 cc distilled water. Subsequently, a quaternary ammonium compound was added, cetyltrimethylammonium bromide (Cetavlon), to reduce surface tension by improving product penetration into the dentin, achieving greater humidification and better antibacterial action of the chelator. This product was named EDTAC9-10.
Citric acid is a weak acid, effective in removing the smear layer, and which has good chemical stability and low cost 11. It is used at 10%, 25% or 50% concentrations with excellent results12-13.
Hard tissue demineralization can be explained based on the solubility product constant principle. When using EDTA to irrigate, it captures the calcium ions (Ca+2) dissolved in the solution, and in turn, other dentin ions dissolve to keep the solubility product constant 8. Each EDTA molecule can bind to a single calcium ion, and when there is balance, no further dissolution occurs.
The use of chelators can erode the dentinal walls, which, according to some authors, could be related to the concentration of the solution, pH, the time the product stays in the canal and/or dental age 6,14-15.
The dentin demineralization caused by EDTA depends on the time the solution remains inside the canal, achieving the greatest effect in the first 5 minutes, after which its action decreases9.
During the chemical reaction, there is an exchange of EDTA hydrogen ions (H+) for Ca+2 ions in the dentin, which reduces the solution’s pH, thus decreasing the chelating action 16.
In 1986, Kennedy, Walker y Gough6 suggested that increased tubule diameter caused by demineralization with chelators depends on the degree of tooth maturation, achieving greater openness in younger teeth that are less mineralized than adult teeth; they suggested implementing greater chelation in the latter to achieve good results.
The objectives of this study were: a) to evaluate the effectiveness of the 17% EDTAC and 10% citric acid prepared formula to remove the smear layer after biomechanical preparation; b) to select the most appropriate chelator exposure time (1 or 3 minutes) to ensure better cleaning of the dentinal walls without eroding them and considering dental age; and c) to evaluate crystal formation when using citric acid.
Materials and methods
The study was approved by the Ethics Committee of the School of Dentistry of Universidad de la República. One hundred and twenty human teeth were used. They had been previously disinfected with 2% glutaraldehyde: 60 from young patients (15-25 years of age) and 60 from adult patients (40 years of age and older). Monoradicular anterior teeth and premolars with fully formed apices were selected. They had been removed due to caries (class I and II), periodontal problems (severe chronic periodontitis) or orthodontics treatment, after having the patient’s informed consent.
Mesiodistal and buccopalatal radiographs were taken, which confirmed that the teeth had one canal, with a curvature of 5° or less according to Schneider’s classification 17, with a radius > 80 mm according to Schäfer’s formula18. The following teeth were discarded: teeth with greater curvatures, those with more than one canal, with resorptions, or previously treated teeth. Chronological age was also assessed to match dental age. A cross-section was made at the neck of the tooth to remove the crown using a refrigerated diamond disc (930 D Jota Switzerland), maintaining a root length of 15 mm. A longitudinal groove was then made on the proximal sides of the root to facilitate tooth section into two halves after canal preparation.
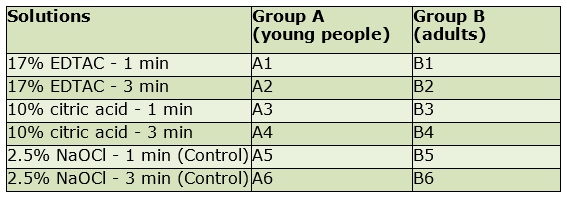
Each subgroup of 60 roots taken from young people (A) and 60 from adults (B) was randomly divided into 6 groups: 4 groups of 12 teeth and 2 control groups of 6 teeth (Table 1).
The working length was determined by penetrating the apical foramen with a K file No. 15 (Dentsply Maillefer Sirona Switzerland) and then 1 mm was substracted. The cervical and middle thirds were expanded with Gates Glidden 3, 2 and 1 burs (Dentsply Maillefer, Ballagues Switzerland) and the rest of the canal with K limes (Dentsply Maillefer Sirona Switzerland) applying the step back technique. The apical third was prepared up to a 35 or 40 file. The young teeth required higher caliber apical preparation (No. 50 to 70) given the canal width. A 5 ml hypodermic syringe with a 25G needle was used for irrigation with 2 ml of 2.5% NaOCl at each instrument change and reassessing constantly. As final irrigation (canal cleansing), it was washed with 2 ml of 2.5% NaOCl, aspirated with syringe and dried with paper cones (Antaeos, VDW Germany). It was then irrigated with 1 ml of the chelator to be tested with an insulin syringe (27G needle). These solutions (prepared at the Biochemistry Laboratory of the School of Dentistry, Universidad de la República) were kept in the canal for 1 or 3 minutes depending on the group, stirring them for 30 seconds with a K15 file (Dentsply Maillefer Sirona Switzerland) so the solution would have greater effectiveness. The chelator was removed by washing with 4 ml of 2.5% NaOCl and dried with paper cones. In all cases, the apical foramen was sealed with resinous wax (Dentorium USA) to prevent irrigant leakage.
The roots were sectioned longitudinally, fitting an instrument into the grooves previously made on the proximal surfaces. The samples were observed in the cervical, middle, and apical thirds using the scanning electron microscope (Jeol JSM 5900 LV Japan) in x1000 and x4000 magnifications, obtaining 360 images at each magnification.
A scale with four levels was created to evaluate the presence of a smear layer:
1.Absence of smear layer: open and clean tubules.
2.A thin smear layer on the surface: some tubules (less than 50%) with smear layer covering the openings.
3.Presence of smear layer on the surface and most tubules closed (over 50%).
4.Thick smear layer; tubules are not distinguishable.
A four-level scale was developed to assess the degree of erosion in the walls of the dentinal tubules:
0.No erosion: all tubules look normal.
1.Incipient erosion of the peritubular dentin appearing on the edges of the tubules.
2.Moderate erosion of peritubular dentin.
3.Severe erosion: the intertubular dentin is destroyed and the tubules may be connected to each other.
Three observers were calibrated (Kappa Test: 0.76 for cleanliness and 0.78 for erosion).
The results were evaluated quantitatively and with two variables with the chi-square test and subsequently, using multivariate models of binary logistic regression.
Results
In the control groups where NaOCl was the sole irrigant, a dense smear layer was observed covering the entire canal surface. In contrast, the groups where EDTAC or citric acid and NaOCl were applied, both supplements were effective in removing the smear layer. As for the degree of cleanliness of the two chelators, there was no significant difference (p= 0.267). The level of cleanliness achieved by chelators (EDTAC and citric acid) according to dental age was higher in the young teeth (p= 0.038 and p= 0.00025) respectively.
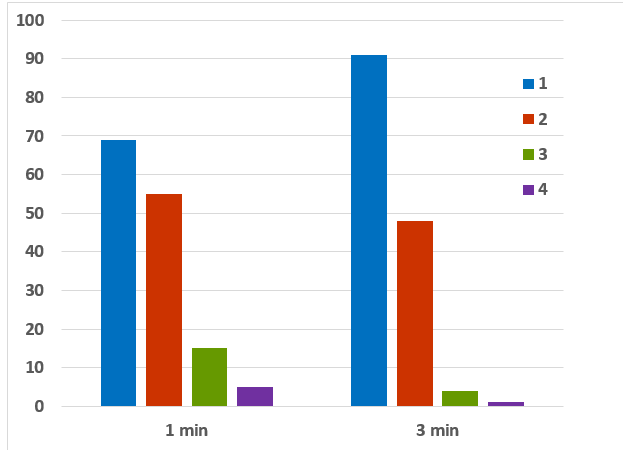
Time was shown to be a dependent variable regarding cleanliness (p= 0.00576) (Chart 1). A multivariate model of binary logistic regression was tested with the cleanliness variable as a dependent variable, and age, time, third and irrigant as independent variables. The model was adjusted (Hosmer and Lemeshow Test, p= 0.601) and showed that age, time, and third of the canal are significantly dependent. The more time, the better cleanliness; the older the tooth, the thicker the smear layer, and the more apical, the less cleanliness. The irrigant did not appear to be significantly dependent.
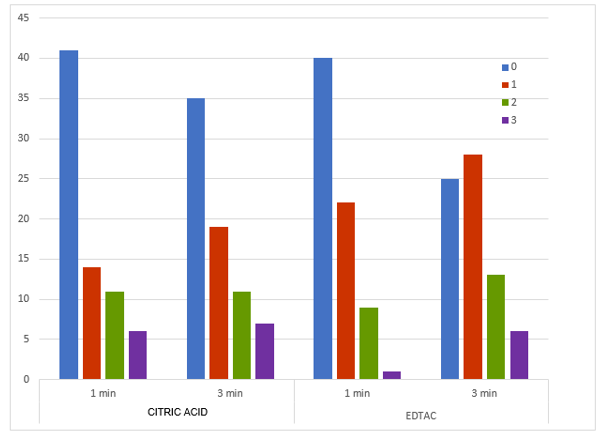
In addition, erosion was evaluated based on the chelator, time of action and dental age. In the samples where EDTAC was applied (groups A1, A2, B1 and B2 in their three thirds) regardless of time and age, 54.9% of them showed erosion. Of the total samples, 15.3% corresponded to level 2 (moderate erosion) and 4.9% to level 3 (severe erosion). Regarding citric acid, 47.2% showed erosion, it being moderate in 15.3% of cases and severe in 9% of samples (Fig. 1). All the groups had most samples with incipient erosion (level 1). Considering the exposure time factor, the EDTAC eroded more at 3 minutes (p= 0.037), while, in the groups using citric acid, erosion was similar at both times (p= 0.727) (Chart 2). Erosion was age independent for both chelators (citric acid: p= 0.984; EDTAC: p= 0.222).
A multivariate model of binary logistic regression was tested taking the erosion variable as dependent, coding the samples as follows: no erosion (0) and erosion (1, 2, 3). The independent variables were time, third, cleanliness and irrigant (Hosmer and Lemeshow Test, p=0.997). Cleanliness was significantly related to erosion so that the greater the cleanliness, the greater the erosion (p= 0.001), and although the irrigant was not significant (p= 0.09), citric acid tended to erode more.
When citric acid was used, crystals were observed in 8.3% of samples (Fig. 2).
Discussion
Final irrigation of the canal with a chelator and NaOCl is essential to reach the right cleanliness and disinfection levels. Both irrigants complement each other by removing the organic and inorganic remnants generated during the instrumentation of the walls, making it possible to clean areas inaccessible with instruments and sealing lateral and accessory canals 19.
Both EDTAC and citric acid achieved smooth, clean walls with open dentinal tubules, unlike control groups, where only NaOCl was used. These findings are consistent with the results of other authors7,11,20.
In 2018, Machado, Garcia, da Silva Neto et al. 21 achieved greater cleanliness in the cervical and middle thirds compared to the apical third using the same chelators, as we did.
When a surfactant (Cetavlon) was added to the EDTA formula, a more effective irrigant solution (EDTAC) was achieved, as shown in the literature 22-23.
The cleanliness achieved in young teeth was greater than in adult teeth with both chelators. This may be because tubule diameter in young teeth is larger, thus improving irrigant penetration. Additionally, the wider canal allows a greater volume of solution to penetrate, so there will be greater dissociation and capture of metal ions, which increases chelation4. Another point to consider is the access the irrigation needle has to the apical third, which is improved in large canals, allowing for better irrigation.
There is no agreement regarding how long the chelator should remain in the canal. This study reported greater efficacy after three minutes, but structural changes in the dentinal walls were noted, which is consistent with other findings 14,24 .
Irrigation with EDTAC caused deterioration at a longer time, while with citric acid, the results were similar in 1 and 3 minutes.
Other studies show clean walls with open tubules, with no changes in the peritubular and intertubular dentin when using EDTA for 1 minute. When the chelator is left for longer, it causes a major change in the canal’s inner surface 14,25-26.
In 2015, Turk, Kaval, y Sen also observed erosion, despite using EDTA and citric acid for 1 minute, at lower concentrations than in this study, 5% and 2.5% respectively27.
Erosion could be observed in groups A and B indicating that age is not a determining factor for demineralization. These results are not consistent with the idea of Kennedy, Walker y Gough 6 who suggested, without conducting a study, that chelators cause greater demineralization in young teeth because they are less mineralized.
In the final irrigation, chelators eliminate the inorganic matter generated during instrumentation, but also act on the dentinal wall by dissolving the hydroxyapatite crystals and exposing the collagen network. Subsequent irrigation with NaOCl would act directly on the collagen, causing excessive surface wear, resulting in excessive opening of the tubules lumen and their joining when the intertubular dentin disappears 2,28. Collagen fibrils are protected by hydroxyapatite crystals, so NaOCl initially causes their denaturation slowly. However, when chelators are used, collagen is exposed and rapidly attacked by it without the protection of the crystals29-31.
Niu, YoshioKa, Kobayashi y Suda 2002 21 and Qian, Shen y Haapasalo 200632 show no erosion when only a chelator is used in final irrigation. In contrast, when NaOCl is used after the chelator, erosion appears.
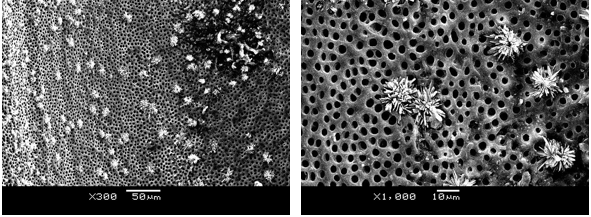
Uninstrumented virgin canal areas were observed in some microphotographs. The presence of unextracted predentin shows an image similar to that of the highly eroded, dentinal tubules with a broad lumen and interconnected ducts (Fig. 3a). These findings are consistent with other studies33-34.
In the uninstrumented areas, NaOCl eliminated the predentin, exposing a globular surface corresponding to the calcospherites (dentin mineralization fronts) and the action of the chelator, left the dentinal tubules uncovered (Fig. 3 b). These observations are described in other papers2,33.
With the use of citric acid, crystals were observed in some samples as described by other authors3,7,11. Its formation could be due to the sudden increase in the pH of the solution, which is initially very low1,8. By coming into contact with some remaining NaOCl in the canal, its pH would rise, causing its eventual precipitation. Prior to irrigation with the chelator, the canal was dried with paper cones to prevent both solutions from coming into contact, but it was not dried again before final irrigation with NaOCl.
During formation, the crystal grows as one layer appears on top of another until a cabbage-like shape is achieved 35. This research shows crystals with a simple structure, with few layers and some in an initial formation stage. To avoid crystal formation, the canal must be dried between each irrigant and/or a final irrigation with distilled water is necessary to remove them if they are still present.
Conclusions
The use of EDTAC or of citric acid with NaOCl was effective in removing the smear layer and the action was similar with both chelators under the conditions of this study. The longer the exposure time, the cleaner condition was achieved, but more erosion was observed.
Erosion appeared in young and adult teeth, so demineralization is not age-dependent.
Crystals were observed in some samples of the citric acid groups.
Recommendations:
Use EDTAC or citric acid for one minute in both young and adult teeth to remove the smear layer without altering the dentinal walls.
When using citric acid, avoid contact with NaOCl at all times or perform a final irrigation with distilled water to remove any crystals formed.
Acknowledgments
To Dr. Enrique Zynemanas, who carefully prepared the chelating solutions at the Biochemistry Laboratory of the School of Dentistry, Universidad de la República.
To Lic. Ramón Alvarez, who performed the Kappa test in this study.
To Lic. Eduardo Cuitiño, for having prepared the statistical analysis.
To Profs. Drs. Ma. Elia Alonso, Irene Lorenzo, Nelly Añaña and Ronell Bologna, who read and corrected the manuscript with great dedication.
To MSc. Ana Laura Reyes from the School of Sciences, as her professionalism and patience allowed the capture and observation of samples with the scanning electron microscope.
REFERENCES
1. Mc Comb D, Smith DC. A preliminary scanning electron microscopic study of root canals after endodontic procedures. J Endod. 1975; 1 (7): 238-242. [ Links ]
2. Baumgartner JC, Mader CL. A Scanning Electron Microscopic Evaluation of Four Root Canal Irrigation Regimens. J Endod. 1987; 13 (4): 147-157. [ Links ]
3. Mader Cl, Baumgartner JC, Peters DD. Scanning Electron Microscopic Investigation of the Smeared Layer on Root Canal Walls. J Endod. 1984; 10 (10): 477- 483. [ Links ]
4. Hülsmann M, Heckendorff M, Lennon A. Chelanting agents in root canal treatment: mode of action and indications for their use. Int Endod J. 2003; 36 (12): 810-830. [ Links ]
5. Orstavik D, Haapasalo M. Desinfection by endodontic irrigants and dressings of experimentally infected dentinal tubules. Endod Dent Traumatol. 1990; 6 (4): 142-149. [ Links ]
6. Kennedy WA, Walker WA, Gough RW. Smear layer removal effects on apical leakage. J Endod. 1986; 12 (1): 21-27. [ Links ]
7. Yamada RS, Armas A, Goldman M, Lin PS. A Scanning Electron Microscopic Comparison of a High Volume Final Flush with Several Irrigating Solutions: Part 3. J Endod 1983; 9(4):137-142 [ Links ]
8. Ostby NB. Chelation in root canal therapy. OdontolTidskr. 1957; 65: 1-11. [ Links ]
9. Von der Fehr FR, Ostby NB. Effect of EDTAC and sulfuric acid on root canal dentin. Oral Surg, Oral Med, Oral Pathol. 1963; 16 (2): 199-205. [ Links ]
10. Giardino L, Estrela C, Generali L, Mohammadi Z, Asgary S. The in vitro Effect of Irrigants with Low Surface Tension on Enterococcus faecalis. Iranian Endod J. 2015; 10 (3): 174-178. [ Links ]
11. Di Lenarda R, Cadenaro M, Sbaizero O. Effectiveness of 1 mol L-1 citric acid and 15% EDTA irrigation on smear layer removal. Int Endod J 2000; 33 (1): 46-52. [ Links ]
12. Haznedaroglu F. Efficacy of various concentrations of citric acid at different pH values for smear layer removal. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003; 96 (3): 340-344. [ Links ]
13. Wayman BE, Kopp WM, Pinero GL, Lazzari EP. Citric and lactic acids as root canal irrigants in vitro. J Endod. 1979; 5(9): 258-265. [ Links ]
14. Torabinejad M, Khademi AA, Babagoli J, Cho Y, Johnson WB, Bozhilov K et al.: A New Solution for the Removal of the Smear Layer. J Endod. 2003; 29 (3): 170-175. [ Links ]
15. Serper A, Calt S. The Demineralizing Effects of EDTA at Different Concentrations and pH. J Endod 2002; 28 (7): 501-502. [ Links ]
16. Seidberg BH, Schilder H. An evaluation of EDTA in endodontics. Oral Surg Oral Med Oral Pathol 1974; 37 (4): 609-620. [ Links ]
17. Schneider SW. A comparison of canal preparations in straight and curved root canals. Oral Surg Oral Med Oral Pathol . 1971; 32 (2): 271-275. [ Links ]
18. Schäfer E, Diez C, Hoppe W, Tepel J. Roentgenographic investigation of frequency and degree of canal curvatures in human permanent teeth. J Endod. 2002; 28(3): 211- 216. [ Links ]
19. Ricucci D, Siqueira JF. Fate of the Tissue in Lateral Canals and Apical Ramifications in Response to Pathologic Conditions and Treatment Procedures. J Endod. 2010; 36 (1): 1-15. [ Links ]
20. Khedmat S, Shokouhinejad N. Comparison of the Efficacy of Three Chelating Agents in Smear Layer Removal. J Endod. 2008; 34 (5):599-602. [ Links ]
21. Machado R, Garcia LDFR, da Silva Neto UX, Cruz Filho AMD, Silva RG, Vansan LP. Evaluation of 17% EDTA and 10% citric acid in smear layer removal and tubular dentin sealer penetration. Microsc Res Tech. 2018; 81 (3): 275-282 [ Links ]
22. Giardino L, Ambu E, Becce C, Rimondini L, Morro M. Surface Tension Comparison of Four Common Root Canal Irrigants and Two New Irrigants Containing Antibiotic. J Endod. 2006; 32(11): 1091-1093. [ Links ]
23. Goldberg F, Abramovich A. Analysis of the effect of EDTAC on the dentinal walls of the root canal. J Endod. 1977; 3 (3): 101-105. [ Links ]
24. Niu W. Yoshioka T. Kobayashi C. Suda H. A scanning electron microscopic study of dentinal erosion by final irrigation with EDTA and NaOCl solutions. Int Endod J. 2002; 35 (11): 934-939. [ Links ]
25. Calt S, Serper A. Time- Dependent Effects of EDTA on Dentin Structures. J Endod 2002: 28 (1): 17-19. [ Links ]
26. Baldasso FER, Roleto L, Silva VDD, Morgental RD, Kopper PMP. Effect of final irrigation protocols on microhardness reduction and erosion of root canal dentin. Braz Oral Res. 2017; 15: 31-40. [ Links ]
27. Turk T, Kaval ME, Sen BH. Evaluation of the smear layer removal and erosive capacity of EDTA, boric acid, citric acid and desy clean solutions: an in vitro study. BMC Oral Health. 2015; 15: 104 [ Links ]
28. Marending M, Paqué F, Fischer J, Zehnder M. Impact of Irrigant Sequence on Mechanical Properties of Human Root Dentin. J Endod. 2007; 33 (11): 1325-1328. [ Links ]
29. Zhang K, Kim YK, Cadenaro M, Bryan TE, Sidow SJ, Loushine RJ, Ling JQ, Pashley DH, Tay FR. Effects of different exposure times and concentrations of sodium hypochlorite/ethylenediaminetetraacetic acid on the structural integrity of mineralized dentin. J Endod. 2010; 36 (1): 105-109 [ Links ]
30. Zhang K, Tay FR, Kim YK, Mitchell JK, Kim JR, Carrilho M, Pashley DH, Ling JQ. The effect of initial irrigation with two different sodium hypochlorite concentrations on the erosion of instrumented radicular dentin. Dent Mater. 2010; 26 (6): 514-523 [ Links ]
31. Tartari T, Bachmann L, Zancan RF, Vivan RR, Duarte MAH, Bramante CM. Analysis of the effects of several decalcifying agents alone and in combination with sodium hypochlorite on the chemical composition of dentine. Int Endod J. 2018; 51 Suppl 1: 42- 54. [ Links ]
32. Qian W, Shen Y, Haapasalo M. Quantitative Analysis of the Effect of Irrigant Solution Sequences on Dentin Erosion. J Endod. 2011; 37 (10): 1437-1441. [ Links ]
33. Haapasalo M, Qian W, Shen Y. Irrigation: beyond the smear layer. Endodontic Topics 2012; 27(1): 35-53. [ Links ]
34. Goldman LB, Goldman M, Kronman JH, Lin PS. The efficacy of several irrigating solutions for endodontics: A scanning electron microscopic study. Oral Surg Oral Med Oral Pathol. 1981; 52 (2): 197-204. [ Links ]
35. Martinelli S, Strehl A, Mesa M. Estudio de la eficacia de diferentes soluciones de EDTA y ácido cítrico en la remoción del barro dentinario. Odontoestomatología. 2012; 14 (19): 52-63. [ Links ]
Authorship contribution 1.Conception and design of study 2.Acquisition of data 3.Data analysis 4.Discussion of results 5.Drafting of the manuscript 6.Approval of the final version of the manuscript GA has contributed in: 2, 3 and 4 LS has contributed in: 2, 3 and 4 SM has contributed in: 1, 2, 3, 4, 5 and 6
Received: December 02, 2018; Accepted: July 15, 2019










 texto em
texto em