Serviços Personalizados
Journal
Artigo
Links relacionados
Compartilhar
Odontoestomatología
versão impressa ISSN 0797-0374versão On-line ISSN 1688-9339
Odontoestomatología vol.21 no.33 Montevideo jun. 2019 Epub 01-Jun-2019
https://doi.org/10.22592/ode2019n33a9
Update
Role of estrogens in the genesis of painful musculoskeletal joint disorders
1Pontificia Universidad Católica de Chile, kvvonbischhoffshause@uc.cl
2Pontificia Universidad Católica de Chile
3Facultad de Medicina Pontificia Universidad Católica. Escuela de Odontología, Internado Intramural de Oclusión y Trastornos Temporomandibulares
Introduction:
The prevalence of Temporomandibular Disorders (TMD) tends to be higher among women. The severity of their symptoms is related to the age of the patients, in the case of women with a peak between 20-40 years.
Objective:
The objective of this study is to carry out a literature review regarding the role of estrogens in joint musculoskeletal disorders.
Method:
The online search engine PubMed was used with the terms MeSH "Temporomandibular Joint Disorders” and “Estrogens” together with the Boolean term “AND”.
Results:
We observed a positive relationship between the levels of estrogen and the presence of muscular and articular disorders, with higher prevalence among women.
Conclusions:
The role of estrogen in muscle and joint disorders is based mostly on correlations between one and the other, but the mechanism by which they would be associated is not entirely clear.
Keywords: temporomandibular joint disorders; estrogens
Introducción:
La prevalencia de los Trastornos Temporomandibulares (TTM), tiende a ser mayor en mujeres. La severidad de su sintomatología está relacionada con la edad de los pacientes, en el caso de las mujeres con un peak entre los 20-40 años.
Objetivo:
El objetivo de este estudio es realizar una revisión bibliográfica de la literatura acerca del rol que podrían tener los estrógenos en los trastornos músculo esqueléticos articulares.
Método:
Se utilizó el buscador online PubMed con loas términos Mesh “Temporomandibular Joint Disorders“ y “Estrogens“ junto con el término bolleano “AND“.
Resultados:
Se aprecia una relación positiva entre los niveles de estrógeno y la presencia trastornos musculares y articulares, siendo en la mujeres mas prevalentes.
Conclusiones:
El rol de los estrógenos en los trastornos musculares y articulares se basan más que nada en correlaciones entre uno y otro, pero el mecanismo por el cual estarían asociados no esta del todo claro.
Palabras clave : Trastornos Temporomandibulares; Estrógeno
Introduction
Temporomandibular Disorders (TMD) are a diverse set of clinical conditions characterized by pain and dysfunction of the masticatory system that may involve masticatory muscles, the temporomandibular joint (TMJ) or related structures1. They are a serious public health issue that affects approximately between 5 and 12% of the population2. They are the second most common cause of chronic painful musculoskeletal conditions and can affect people’s daily life, psychosocial functioning and quality of life (2-3. It is characterized by a triad of symptoms, including joint noise, pain and limited mandibular movements4.
The incidence of these disorders tends to be higher in women than in men and moreover, the severity of their symptoms is related to the age of the patients (1. The prevalence of TMD before adolescence is low and seems not to be different for boys and girls. They tend to occur after puberty with a peak between 20-40 years for women, and to a lesser extent in children and the elderly1. Additionally, prevalence is lower for postmenopausal women than for women of reproductive age. This epidemiological variation hints at the role female reproductive hormones, especially estrogens, may have in the genesis of TMD1,5.
The objective of this study is to carry out a literature review regarding the potential role of estrogens in joint musculoskeletal disorders.
Materials and methods
The online search engine PubMed was used for the literature review with the MeSH terms “Temporomandibular joint disorders” and “Estrogens” together with the Boolean term “AND”.
The inclusion criteria was to include studies related to the subject matter of this review, research articles, reviews and clinical cases with studies in humans, which may be found and whose full text was accessible. Articles published in English or Spanish were included.
The exclusion criteria was to exclude animal studies and studies withdrawn by the original journal.
Results
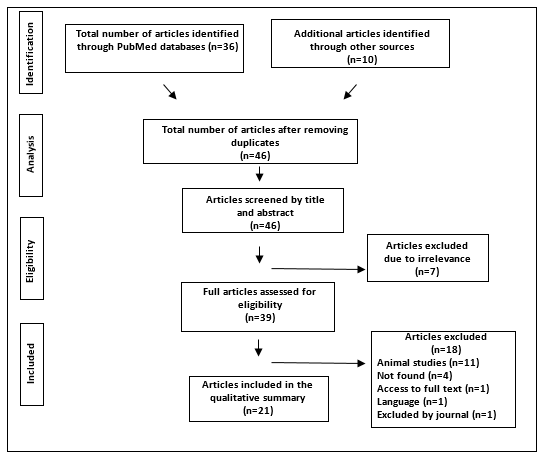
Thirty-six studies were obtained. Ten studies identified through searches in other sources were also added to this search. Forty-six articles were collected in the entire search, seven of which were excluded for not being related to the subject matter. Out of all the articles selected, those which did not meet the inclusion criteria were excluded, 11 of them because they were about animal studies, 4 could not be found, 1 because the full text could not be accessed, 1 due to the language and 1 was withdrawn by the journal after publication. The total number of studies analyzed in this review was 21, as shown in Figure 1.
Menstrual cycle
The menstrual cycle is a carefully orchestrated sequence of hormonal events which can be divided into a follicular phase and a luteal phase. Estrogen secretion gradually increases during the first part of the follicular phase and then exponentially in the days before ovulation. Ovulation occurs approximately 10 to 12 hours after the LH peak. The luteal phase begins after ovulation, when estrogen levels begin to decrease to then gradually increase during the beginning and middle of the luteal phase. Estrogen then drops again during the late luteal phase right before menstruation5-6 as shown in Figure 2.
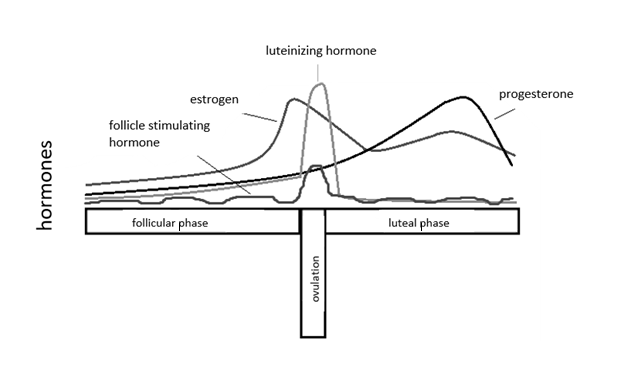
Figure 2: Chart showing the variation in hormone levels during the different stages of the menstrual cycle
Women taking oral contraceptives experience a fall of estrogen at the end of the cycle when they stop taking the active medication, and have relatively low levels of estrogen during menstruation. However, they do not experience the fluctuations of estrogen levels associated with ovulation5.
The physiology of gonadal steroid hormones
Ovaries primarily produce two types of hormones: estrogens, also referred to as estradiol, estriol or estrone, and progestin (progesterone). Testosterone is a precursor of estradiol, so ovaries also produce testosterone. When menopause occurs, there are changes in the endogenous hormone balance with a dramatic decline in estrogen production in the ovaries7. This review will mainly focus on the role estrogen may play in the development of TMD.
Presence of estrogen receptors
The biological activity of estrogens begins when they bind to their estrogen receptors. Three types of estrogen receptors have been identified to this date: two nuclear (ERα and ERβ) and one plasma receptor (GPER1)8. Immunohistochemistry using specific antibodies has revealed that ERα is widely distributed in various tissues, whereas the definitive distribution of ERβ is unclear9.
Despite this, there is still controversy regarding the presence of these receptors in TMJ, as there are many conflicting studies on the presence of this kind of receptors. Abubaker et al. found estrogen receptors in the human TMJ disc and reported that women with TMJ symptoms are five times more likely to have intracapsular estrogen receptors than women without TMJ symptoms10. A possible explanation for this may be that the decline in circulating estradiol lead to an overregulation of estrogen receptors in joint tissues11. Conversely, Campbell et al. found no estrogen receptors in the TMJ12-13. Henry et al. conducted a study to determine the presence of estrogen receptors in the retrodiscal zone of the TMJ. Their research found no cases of receptors in the retrodiscal zone14. The presence of estrogen receptors in other structures in the TMJ has also been assessed, and Galal et al. found estrogen receptors in synovial fibroblasts and chondrocytes4.
Estrogens and temporomandibular disorders
The female:male prevalence ratio for temporomandibular disorders is nearly 2:1 in the general population, whereas in the clinical population it can be 8:19. The neurobiological basis for this higher prevalence is unknown and likely involves peripheral as well as central mechanisms9.
Among the factors that could account for this significant difference between both sexes, there is evidence supporting the significant role of estrogens in temporomandibular disorders:
The expression of temporomandibular disorders follows a pattern consistent with the development and regression of the ovarian production of estrogens
The prevalence and severity of temporomandibular disorders in females is similar to males prior to puberty, when estrogen production is low, rises during the reproductive years, with a peak between 20-40 years for women, and diminishes after menopause, following a prevalence pattern that varies with the different stages of life and which may be related to estrogen production1,5.
Variations in estrogen production over the menstrual cycle and during pregnancy
The intensity of musculoskeletal pain associated with TMD varies systematically across the menstrual cycle5 and cyclical fluctuations of ovarian hormones during the menstrual cycle have a significant effect on pain.
Women with TMD experience the highest levels of pain when estrogen levels are low or fluctuating5. For women with TMD who have regular menstrual cycles, facial pain increased with the fall of estrogen that takes place in the late luteal phase and reached its peak during menstruation, when estrogen reaches its lowest level5,12. Vilanova et al. obtained similar results showing an increase of pain during the luteal and menstrual phases3. A study conducted by Martin et al. found that of a total of 19 studies, 7 showed an increase in pain reactivity during the late luteal phase or the early follicular phase, 5 studies showed an increase in the pain response during the late follicular phase and the early luteal phase, and 6 studies found no difference6. Finally, women show higher tolerance to pain during the follicular phase than during the luteal phase15, when estrogen levels are higher. During reproductive years, women with TMD experienced more pain during perimenstrual periods, although women who did not take oral contraceptives had an additional peak of pain during the periovulatory periods15.
There are also studies that link the presence of TMD to higher concentrations of estradiol1,16. This link has also been reported in other studies in which serum levels of estradiol during the luteal phase of the female menstrual cycle were significantly higher in patients with TMD than in healthy controls12. Similar results were found when comparing the levels of estradiol in the TMJ synovial fluid of patients with TMD12.
During pregnancy, women with musculoskeletal TMD experience an improvement in pain during pregnancy, returning to baseline pain levels one year postpartum. The improvement in pain is likely associated with the dramatic hormonal changes that occur during pregnancy. Reported musculoskeletal orofacial pain diminished significantly during the second or third trimester of pregnancy17). This is complemented by a study conducted by Ivkovic et al., in which pain decreased with the progress of pregnancy and was the lowest at the 36th week of pregnancy18).
Exogen estrogen
Synthetic estrogen is an active analogue of 17β-estradiol, but it does not have the same effect on target organs or on known estrogen receptors (ERα or ERβ) as 17β-estradiol. In addition, exogenous estrogen suppresses the natural production of 17β-estradiol11. The action of exogenous estrogen is to suppress the production and availability of natural 17β-estradiol, resulting in an increase in osteoclastic activity and in the production of inflammatory cytokines11.
Use of oral contraceptives by women of reproductive age or hormone replacement therapy is associated with a higher prevalence of TMD3. Abubaker established a link between the higher use of exogenous estrogen in women and TMD1,10. It was also established that excessive ovarian hormones can put a woman at risk of developing a TMD1. The results obtained by Hatch et al., however, do not support this correlation. The prevalence and severity of TMD signs and symptoms were almost identical in women taking estrogens or not, both for muscle and joint disorders19.
As for estrogen replacement therapy, it was found that it increases temporomandibular disorders after menopause. The odds of being a TMD case were approximately 30% higher among women receiving exogenous estrogen compared to those not exposed1. There is an increased risk of suffering a TMD for women using postmenopausal hormones20. A study in which Ivkovic et al. compared different control groups (women with normal menstrual cycles, pregnant women and postmenopausal women receiving estrogen replacement therapy) concluded that postmenopausal women reported the highest pain intensity18.
It was also found that for women with TMD using oral contraceptives, orofacial pain intensity levels begin to rise toward the end of the cycle and peak during the first days of menstruation5. However, a study conducted by McFarlane et al. did not show a higher risk of orofacial pain in patients taking oral contraceptives20. Vilanova et al. concluded that patients taking oral contraceptives also experienced episodes of pain during hormone-free intervals, which also increased during the luteal and menstrual phases3. Dao et al. showed greater variability in the severity of myofascial pain during the day in women with normal cycles than those receiving oral contraceptives6,21. Finally, it was also found that premenopausal women taking contraceptive pills are given 20% more referrals for TMD care than those who do not use oral contraceptives22.
Effect of estrogen on inflammation
Steroid hormones, especially estrogens, act on their receptors (alpha and beta) in the peripheral and central nervous systems, which has effects on the inflammatory process and on the central transmission of pain. For example, estrogens can act directly on monocytes and macrophages, to regulate the production of cytokines (e.g., IL-1, IL-6, FNT-α). IL-1b and IL-6 are present in the TMJ synovial fluid during inflammation, and IL-1 and TNF-α promote cartilage resorption, inhibit proteoglycan and promote inflammation in most TMJ structures. Additionally, monocytes/macrophages are immune cells present in synovial tissues and are frequently recruited in synovial inflammation, suggesting that most IL-1 and TNF-α released in the joint originate from said immune cells23. Galal et al. detected the expression of M-CSF (acidic glycoproteins which regulate the survival, proliferation and differentiation of hemopoietic progenitor cells into mature macrophages and/or granulocytes) and c-fms in synovial fibroblasts and chondrocytes, showing an increase of these genes with estrogen. This increase in macrophages could be a contributing factor in the increased inflammation and tendency of females to suffer more from TMD4.
Finally, TMD, especially those associated with trauma, disc alterations or inflammatory arthritis, include an inflammatory component. Therefore, estrogens, as well as other sex hormones, can have an important role in the severity of pain and predisposition to TMD23.
Effects of estrogen on cartilage
Some studies have suggested that estrogen plays a role in the development of osteoarthritis due to the presence of estrogen receptors types α and β in chondrocytes1,4.
Effect of estrogen on the condyle and bone metabolism:
One of the factors associated with the onset of temporomandibular joint disorders, in particular, condylar bone lysis, is the decline in estrogens because the 17β-estradiol form has various functions that protect joints24,25.
First, it has a releasing effect on osteoprotegerin (OPG), a protector of inflammatory mechanisms. Therefore, women with a lack of this circulating hormone have increased inflammatory cytokines, inhibiting the formation of new bone, consequently promoting condylar resorption as well as a decrease in bone density11. Estrogen is an important modulator or bone metabolism. It has an effect on osteoblast differentiation, reducing cell proliferation and differentially regulating the expression of the extracellular matrix, which also explains molecular mechanisms of bone growth and remodeling by estrogens. Estrogen increases inflammation and decreases bone resorption11.
Secondly, 17β-estradiol plays a role in metalloproteinase (MMPs) signaling. MMPs are responsible for bone and cartilage destruction and are produced by osteoclast26, and 17β-estradiol is involved in osteoclast differentiation and activation, as well as in metalloproteinase transcription11,26.
Progressive condylar resorption (PCR) is a condition in which low levels of estrogens have been found and which has a higher prevalence in women (25. According to Gunson, it is influenced by the presence of three group of factors: occlusion, local and systemic factors11. The purpose of the study was to evaluate the influence of sex hormones on condylar resorption, and 96% of the women with PCR evaluated were found to have low levels of 17β-estradiol or a history of extremely irregular menstrual cycles, 62% of them met both criteria. 17β-estradiol deficiency is described as a systemic factor for PCR. Other studies also support the idea that sex hormones, especially estrogens, play a role in bone, cartilage and disc tissue regulation27-28. To reinforce the above, an MMP elevation has been found in patients with severe PCR, which would initiate the degradation process for the mandibular condyle's extracellular matrix11.
Effect on other TMJ structures
Estrogen receptors type α and β have been found in synovial fibroblasts or type B cells, indicating that they may have an effect on the synovial membrane1,4.
Estrogen may strengthen the effect of relaxin, which causes muscle relaxation, increasing the expression of specific tissue-degrading enzymes. Laxity of the TMJ is thought to play a role in the development of TMD and suggests a mechanism by which the combined effect of relaxin and estrogen may predispose women to TMD(1.
Estrogen has also been found to induce a significant increase in myoblast growth1.
Proposed mechanisms
Reproductive hormones may act centrally to influence pain neurotransmission.
1.Estrogen receptors in trigeminal neurons modulate nociceptive responses through effects on galanin and neuropeptide Y. Variations in neuropeptide content in trigeminal neurons across the natural reproductive cycle may contribute to an increase in painful episodes at particular phases of the menstrual cycle. Nitric oxide (NO) is a cellular messenger derived from the vascular endothelium and has important biologic roles, including in inflammation and the central processing of pain. Studies in humans have shown that the acute or chronic administration of estrogens can induce the release of NO, and the increase of NO concentration in the TMJ fluid is related to the onset of pain1,21,29.
2.Estrogen may regulate pain by modulating pain levels. It has been demonstrated that pain levels in patients taking oral contraceptives remain positive during the hormonal cycle, whereas those who do not use this medication periods of pain alternate with pain-free periods1,21.
Locally synthesized estrogen: Recently, locally synthesized estrogens have been found and proved to contribute greatly to the function of cartilage. We hypothesize that estrogens synthesized locally in condylar cartilage have a strong effect on the development of TMD12
3.The presence of receptors in peripheral and central tissues may indicate that estrogen can modulate pain and, depending on the type of pain signal, estrogen can act as a pronociceptive or an antinociceptive agent. In physiological pain, estrogen seems to decrease pain while, in inflammatory pain, estrogen acts differently depending on the inflammation type. In acute inflammatory pain caused by formalin or ATP injection, estrogen has an antinociceptive effect. The pronociceptive effect of this hormone is seen in the chronic inflammatory pain model with complete Freund's adjuvant (CFA) or carrageen injection8.
4.Genetic polymorphisms of estrogen receptors alter the susceptibility of women to temporomandibular disorders. Ribeiro Dasilva et al. analyze two receptor-alpha polymorphisms and their relation to TMD. The results show that the presence of two estrogen receptor-alpha gene polymorphisms, PVULL (T-397 C) and Xba I (A-351G) in patients who also have arthritis is associated with higher susceptibility to pain in the TMJ23. As a result, a genetic variation at the ERα could lead to significant modifications in the physiological role of estrogen and consequently in TMD23 Therefore, it is believed that this genotype can affect the level or function of estrogen receptors alpha or the activity of estrogens in inflammatory mediators23. Moreover, in a study conducted by Quinelato et al. it was discovered that the TT (timin/timin) genotype for the ESR1 (rs2273206) gene was strongly associated with the risk of developing muscle TMD and temporomandibular joint pain, and for the ESRRB (rs1676303) gene, an association was observed between the CC (cytosine/cytosine) genotype and the presence of articular TMD associated with other chronic arthralgia30. Despite this, a study conducted by Kim31 could not demonstrate a correlation between the risk of TMD and different polymorphisms of the estrogen receptor-alpha gene32.
Discussion
The role that estrogens may play in TMD is not entirely clear; there are many hypotheses concerning this, but none of them have a clear scientific basis and most yield contradictory results. Regarding anatomy, studies on the presence or absence of estrogen receptors in the different TMJ structures are conflicting. Though Abubaker et al. and Galal et al. found estrogen receptors in joint discs10 and in synovial fibroblasts and chondrocytes, Campbell et al. found no estrogen receptors in the TMJ12-13. Henry et al. were not able to find receptors in the retrodiscal zone either14, therefore, there are doubts on whether they are present in all women or not.
We also found contradictory results on estrogen variations during the menstrual cycle. Although the majority supports the presence of episodes with more symptoms when estrogen levels are low or fluctuating3,5,12,15, there are other studies which find a correlation between these episodes and the times when estrogen levels are highest1,12, therefore, we were not able to draw a clear conclusion on this matter.
Regarding the role exogenous estrogen in oral contraceptives and hormone replacement therapy may have, although there are studies that establish a connection between the use of oral contraceptives in women of reproductive age or hormone replacement therapies and a higher prevalence of TMD1,3,10,22, other studies have obtained different results finding no relation to exogenous estrogens18-19. This leads us to think that the causal relationship with estrogen is not entirely clear; it is based more on coincidences than on a well-established physiological mechanism.
The possible role of estrogens in progressive condylar resorption has also been assessed. Low concentrations of estrogen may make it lose its protective role in bone metabolism, increasing inflammation and promoting condylar resorption11. We found no studies with any different evidence regarding the role of estrogens in PCR, which introduced a possible bias, since almost all studies were by the same author11,25-26.
Finally, an effect of estrogen has been observed in other TMJ structures, but this is only mentioned by one author1 and we found no other studies on this subject, therefore, results are neither comparable nor conclusive. As for the effect on cartilage, it was only associated with the presence of estrogen receptors in chondrocytes1,4, which also does not provide strong evidence of its possible effect on these structures.
Conclusion
Estrogens may have a significant effect on the genesis and the modulation of pain in painful conditions such as TMD. Understanding the mechanism by which estrogens reduce or increase pain will enable us to better apply measures to prevent and treat this mechanism, therefore, more studies are necessary to understand the physiological mechanism behind the relationship between estrogens and temporomandibular disorders.
Most of the results are mainly based on correlations that may be connected to the reason why temporomandibular disorders are more common in women rather than on scientific evidence.
In addition, the methodology for these kinds of studies is very complicated, because it is very difficult to identify the precise time of ovulation and the phases of the menstrual cycle. We also found other limitations among the studies included in the review and that is that most lack a clear diagnosis for temporomandibular disorders, considering each has a different origin and treatment.
We conclude that more studies with a clearer methodology are necessary in order to evaluate the actual role that estrogens may play in the genesis or perpetuation of these disorders
Referencias
1. Wang J, Chao Y, Wan Q, Zhu Z. The possible role of estrogen in the incidence of temporomandibular disorders. Med Hypotheses. 2008;71: 564-7 [ Links ]
2. Schiffman E, Ohrbach R, Truelove E, Look J, Anderson G, Goulet JP, List T, Svensson P, Gonzalez Y, Lobbezoo F, Michelotti A, Brooks SL, Ceusters W, Drangsholt M, Ettlin D, Gaul C, Goldberg LJ, Haythornthwaite JA, Hollender L, Jensen R, John MT, De Laat A, de Leeuw R, Maixner W, van der Meulen M, Murray GM, Nixdorf DR, Palla S, Petersson A, Pionchon P, Smith B, Visscher CM, Zakrzewska J, Dworkin SF; International RDC/TMD Consortium Network, International association for Dental Research; Orofacial Pain Special Interest Group, International Association for the Study of Pain. Diagnostic Criteria for Temporomandibular Disorders (DC/TMD) for Clinical and Research Applications: Recommendations of the International RDC/TMD Consortium Network and Orofacial Pain Special Interest Group. J Oral facial pain headache. 2014; 28 (1): 6-27. [ Links ]
3. Vilanova LS, Gonçalves TM, Meirelles L, Garcia RC. Hormonal fluctuations intensify temporomandibular disorder pain without impairing masticatory function. Int J Prosthodont. 2015;28 (1): 72-4 [ Links ]
4. Galal N, El Beialy W, Deyama Y, Yoshimura Y, Yoshikawa T, Suzuki K, Totsuka Y. Effect of estrogen on bone resorption and inflammation in the temporomandibular joint cellular elements. Int J Mol Med. 2008;21:785-790 [ Links ]
5. LeResche L, Mancl L, Sherman JJ, et al. Changes in temporomandibular pain and other symptoms across the menstrual cycle. Pain. 2003; 106:253-261 [ Links ]
6. Martin VT. Ovarian hormones and pain response: a review of clinical and basic science studies. Gend Med 2009; 6(Suppl 2):168-92 [ Links ]
7. Craft RM, Mogil JS, Aloisi AM. Sex differences in pain and analgesia: the role of gonadal hormones. Eur. J. Pain. 2004; 8: 397-411. [ Links ]
8. Berger M, Szalewski L, Bakalczuk M, Bakalczuk G, Bakalczuk S and Szkutnik J: Association between estrogen levels and temporomandibular disorders: a systematic literature review. Przegl Menopauz.2015; 14:260-270. [ Links ]
9. Bereiter DA, Okamoto K. Neurobiology of estrogen status in deep craniofacial pain. Int Rev Neurobiol. 2011; 97: 251-84. [ Links ]
10. Abubaker AO, Raslan WF, Sotereanos GC. Estrogen and proges- terone receptors in temporomandibular joint discs of symptomatic and asymptomatic persons: a preliminary study. J Oral Maxillofac Surg 1993; 51: 1096-100 [ Links ]
11. Gunson MJ, Arnett GW, Formby B. Oral contraceptive pill use and abnormal menstrual cycles in women with severe condylar resorption: A case for low serum 17 -estradiol as a major factor in progressive condylar resorption. Am J Orthod Dentofacial Orthop. 2009; 136:772- 779. [ Links ]
12. Yu S1, Xing X, Liang S, Ma Z, Li F, Wang M, Li Y. Locally synthesized estrogen plays an important role in the development of TMD. Med Hypotheses. 2009;72(6): 720-722 [ Links ]
13. Campbell JH, Courey MS, Bourne P, Odziemiec C. Estrogen receptor analysis of human temporomandibular disc. J Oral Maxillofac Surg 1993;51(10):1101-5. [ Links ]
14. Henry CH, Tull GT, Whittum-Hudson JA, Wolford LM. Analysis of estrogen binding sites of the posterior ligament of the human TMJ. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008; 105(6):698-701. [ Links ]
15. Craft RM. Modulation of pain by estrogens. Pain. 2007;132(Suppl 1):S3-12 [ Links ]
16. Dworkin SF, Huggins KH, LeResche L, Von Korff M, Howard J, Truelove E, Sommers E. Epidemiology of signs and symptoms in temporomandibular disorders: clinical signs in cases and controls. J Am Dent Assoc. 1990; 120 (3):273-81 [ Links ]
17. LeResche L, Sherman JJ, Huggins K, Saunders K, Mancl LA, Lentz G, Dworkin SF . Musculoskeletal orofacial pain and other signs and symptoms of temporomandibular disorders during pregnancy: a prospective study. J Orofac Pain. 2005; 19 (3):193-201. [ Links ]
18. Ivkovic N, Racic M, Lecic R, Bozovic D, Kulic M. Relationship Between Symptoms of Temporomandibular Disorders and Estrogen Levels in Women With Different Menstrual StatusJ Oral Facial Pain Headache. 2018 21;32 (2): 151-158 [ Links ]
19. Hatch JP, Rugh JD, Sakai S, et al. Is use of exogenous estrogen associated with temporomandibular signs and symptoms? J Am Dent Assoc. 2001;132:319-326 [ Links ]
20. Macfarlane T. V., Blinkhorn A. S., Davies R. M., Kincey J., Worthington H. V. Association between female hormonal factors and oro-facial pain: study in the community. Pain. 2002;97(1-2):5-10. [ Links ]
21. Dao TT, Knight K, Ton-That V. Modulation of myofascial pain by the reproductive hormones: A preliminary report. J Prosthet Dent 1998;79:663-70. [ Links ]
22. LeResche L, Saunders K, Von Korff MR, Barlow W, Dworkin SF. Use of exogenous hormones and risk of temporomandibular disorder pain. Pain. 1997;69(1-2):153-60. [ Links ]
23. Ribeiro-Dasilva MC, Peres Line SR, Leme Godoy dos Santos MC, Arthuri MT, Hou W, Fillingim RB, Rizzatti Barbosa CM. Estrogen receptor-alpha polymorphisms and predisposition to TMJ disorder. J Pain. 2009; 10 (5): 527-33. [ Links ]
24. Arnett, G. Gunson, M. Risk Factors in the Initiation of Condylar Resorption. Semin Orthod. 2013; 2: 81-8. [ Links ]
25. Gunson MJ, Arnett GW, Milam SB: Pathophysiology and pharmacologic control of osseous mandibular condylar resorption. J Oral Maxillofac Surg 2012; 70:1918-1934 [ Links ]
26. Gunson MJ, Arnett GW. Condylar resorption, matrix metalloproteinases, and tetracyclines. RWISOJ. 2010;2:37-44. [ Links ]
27. Kapila S, Wang W, Uston K. Matrix metalloproteinase induction by relaxin causes cartilage matrix degradation in target synovial joints. Ann N Y Acad Sci. 2009;1160:322-28 [ Links ]
28. Hajati AK, Alstergren P, Nasstrom K, Bratt J, Kopp S. Endogenous glutamate in associ-ation with in?ammatory and hormonal factors modulates bone tissue resorption of thetemporomandibular joint in patients with early rheumatoid arthritis. J Oral MaxillofacSurg. 2009; 67:1895-903. [ Links ]
29. Puri J, Hutchins B, Bellinger LL, Kramer PR. Estrogen and inflammation modulate estrogen receptor alpha expression in specific tissues of the temporomandibular joint. Reprod Biol Endocrinol. 2009; 7: 155. [ Links ]
30. Quinelato V, Bonato LL, Vieira AR, Granjeiro JM, Tesch R, Casado PL. Association Between Polymorphisms in the Genes of Estrogen Receptors and the Presence of Temporomandibular Disorders and Chronic Arthralgia.. J Oral Maxillofac Surg. 2018; 76 (2): 314.e1-314.e9 [ Links ]
31. Kim BS, Kim YK, Yun PY, Lee E, Bae J. The effects of estrogen receptor a polymorphism on the prevalence of symptomatic temporomandibular disorders. J Oral Maxillofac Surg. 2010; 68: 2975-9 [ Links ]
32. Melis M, Di Giosia M. The role of genetic factors in the etiology of temporomandibular disorders: a review. Cranio. 2016; 34 (1): 43-51. [ Links ]
Received: September 05, 2018; Accepted: December 12, 2018










 texto em
texto em