PAUTAS
Uso del óxido nítrico en pediatría
Dras. Amanda Menchaca 1, Alicia García 2
1. Profesora Directora de UCIN Facultad de Medicina UDELAR. Centro Hospitalario Pereira Rossell
2. Profesora Adjunta de UCIN Facultad de Medicina UDELAR. Centro Hospitalario Pereira Rossell
Introducción
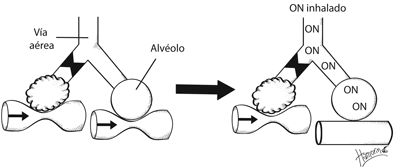
Es el vasodilatador fisiológico más rápido. Se sintetiza a partir L-arginina a través de la óxido nítrico sintetasa (calcio dependiente). Activa la guanilato ciclasa y produce monofosfato de guanosina cíclico que dilata el músculo liso.
]]> Por vía inhalada tiene una vida media muy corta.Se metaboliza en el lecho vascular pulmonar sin llegar a la circulación sistémica, lo que explica su efecto pulmonar selectivo.
Efectos
Indicaciones
Recomendaciones
Si luego de optimizar la ventilación mecánica convencional persiste:
Índice de oxigenación = 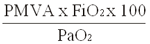
(PMVA: presión media de vía aérea)
PMVA= 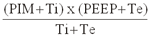
(PIM: presión inspiratoria máxima; Ti: tiempo
inspiratorio; PEEP: presión positiva al final de la
espiración; Te: tiempo espiratorio)
Aplicar el siguiente algoritmo:
1) Criterios clínicos ® Sí ® conexión al sistema.
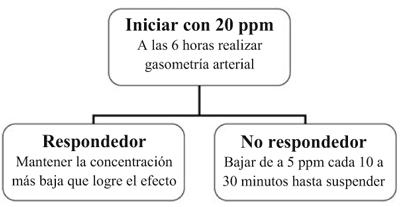
2) Criterio de mejoría o “respondedor”: descenso de Fio2 y/o IO en un 20%.
3) El efecto se ve a los 5 minutos.
4) En patología pulmonar los efectos se logran entre 5 a 20 ppm.
5) nLa concentración óptima debe individualizarse en cada paciente.
6) La duración promedio del tratamiento será entre 24 a 96 horas.
]]> 7) No interrumpir en forma brusca pues produce efecto rebote con hipertensión pulmonar e hipoxemia grave.Controles
Durante la administración de ON es necesaria la medición continua de la concentración de ON y dióxido de nitrógeno (NO2) en el circuito respiratorio
Efectos secundarios
El ON no produce efectos secundarios a las concentraciones habitualmente utilizadas en la práctica clínica, siempre que se eviten concentraciones muy elevadas o la retirada brusca de su administración. No se ha demostrado riesgo de contaminación ambiental ni entre el personal sanitario
1. Gattinoni L, Tognoni G, Pesenti A, Taccone P, Mascheroni D, Labarta V, et al. Prone-Supine Study Group. Effect of prone positioning on the survival of patients with acute respiratory failure. N Engl J Med 2001; 345(8): 568-73.
2. Staudinger T, Kofler J, Mullner M, Locker GJ, Laczika K, Knapp S, et al. Comparison of prone positioning and continuous rotation of patients with adult respiratory distress syndrome: results of a pilot study. Crit Care Med 2001; 29(1): 51-6.
3. Lundin S, Mang H, Smithies M, Stenqvist O, Frostell C. Inhalation of nitric oxide in acute lung injury: results of a European multicentre study. The European Study Group of Inhaled Nitric Oxide. Intens Care Med 1999 25(9):911-9.
4. Dellinger RP, Zimmerman JL, Taylor RW, Straube RC, Hauser DL, Criner GJ, et al. Effects of inhaled nitric oxide in patients with acute respiratory distress syndrome: results of a randomized phase II trial. Inhaled Nitric Oxide in ARDS Study Group. Crit Care Med 1998; 26(1): 15-23.
5. Troncy E, Collet JP, Shapiro S, Guimond JG, Blair L, Ducruet T, et al. Inhaled nitric oxide in acute respiratory distress syndrome: a pilot randomized controlled study. Am J Respir Crit Care Med 1998; 157(5 Pt 1): 1483-8.
6. Sokol J, Jacobs SE, Bohn D. Óxido nítrico inhalado para la insuficiencia respiratoria aguda hipoxémica en niños y adultos (revisión Cochrane traducida). En: La Biblioteca Cochrane Plus, número 4, 2008. Oxford : Update Software Ltd.Disponible en: http://www.update-software.com ]]>