Artículo original
Utilidad del score SAMe-TT2R2 en el control de la anticoagulación oral con warfarina en pacientes con fibrilación auricular no valvular
Dres. Andreina Gómez, Stephania Peixoto, Lic. Enf. María Azcúnaga,
Dres. Natalia Lluberas, Gabriela Silvera, Pablo Álvarez, Gabriela Ormaechea, Ricardo Lluberas
Centro Cardiovascular Universitario y Unidad Multidisciplinaria de Insuficiencia Cardíaca. Hospital de Clínicas. Montevideo, Uruguay.
Correo electrónico: andreinagomezeli@gmail.com
Recibido Mar 1, 2016; aceptado Set 21, 2016
Antecedentes: la prevención de eventos tromoboembólicos mediante anticoagulación oral es uno de los principales objetivos en el tratamiento de la fibrilación auricular (FA) no valvular, pudiendo utilizarse tanto fármacos antivitamina K (AVK), como warfarina y anticoagulantes orales directos (ACOD). La warfarina ofrece su mayor eficacia y seguridad cuando el porcentaje de tiempo en rango terapéutico (TTR) es mayor de 65%-70%. El score SAMe-TT2R2 fue desarrollado como herramienta para intentar predecir la respuesta al tratamiento anticoagulante con fármacos AVK. Los pacientes con un puntaje favorable (0-1 punto) tendrían una buena respuesta al tratamiento y por lo tanto un TTR adecuado, mientras que un puntaje desfavorable (2 puntos) permitiría predecir un TTR inadecuado, identificando pacientes que requieren intervenciones adicionales para optimizar la calidad de anticoagulación o que serían mejores candidatos a ACOD.
Objetivo: evaluar la utilidad del score SAMe-TT2R2 en el control de la anticoagulación oral con warfarina en pacientes portadores de FA no valvular.
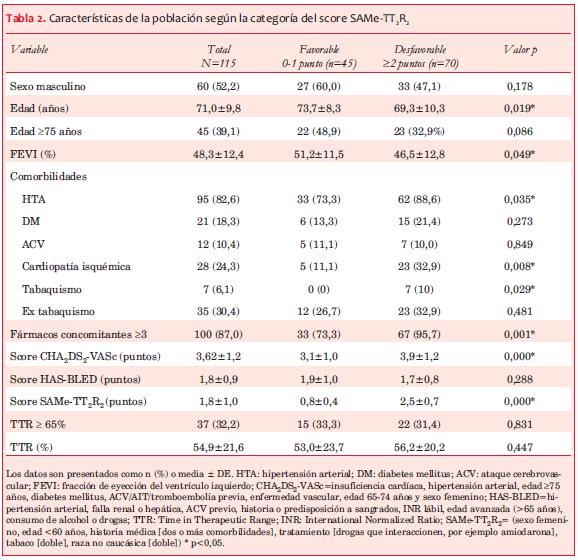
Método: estudio retrospectivo de 115 pacientes ambulatorios con FA no valvular, anticoagulados con warfarina entre el 1° de junio de 2012 y el 31 de junio de 2014. Las variables analizadas fueron: edad, sexo, fracción de eyección del ventrículo izquierdo (FEVI), comorbilidades, fármacos concomitantes, score CHA2DS2-VASc y HAS-BLED. Se calculó el TTR individual mediante método de Rosendaal y se calculó el score SAMe-TT2R2. Se utilizó el test de t para comparación de medias y el test de chi2 para análisis de variables categóricas. Se consideró significativo un valor p<0,05.
Resultados: la media de edad fue de 71,0±9,8 años, sexo masculino 52,2%. Comorbilidades asociadas fueron: hipertensión arterial (HTA) 82,6%, cardiopatía isquémica 24,3%, diabetes mellitus 18,3%, ataque cerebrovascular previo 10,4%, tabaquismo 6,1% y ex tabaquista 30,4%, consumo concomitante de tres o más fármacos 87,0%. La media de FEVI fue de 48,3±12,4%, score CHA2DS2-VASc 3,6±1,2 puntos y score HAS-BLED 1,8±0,9 puntos. La media de TTR calculada fue de 54,9±21,6% y solamente 37 pacientes (32,2%) tuvieron un TTR 65%. El score SAMe-TT2R2 tuvo una media de 1,8±1,0 puntos, 45 pacientes (39,1%) tuvieron un puntaje favorable a warfarina (0-1 punto) y 70 pacientes (60,9%) un puntaje desfavorable (2 puntos). No hubo diferencia significativa en la media de TTR según la categoría de SAMe-TT2R2 (53,0±23,7% vs 56,2±20,2%, p=0,447). Tampoco se encontró asociación entre un score SAMe-TT2R2 favorable a warfarina y un TTR 65% (33,3% vs 31,4%, p=0,831).
Conclusión: en la población estudiada no hubo diferencia en la calidad de la anticoagulación oral con warfarina entre las categorías (favorable y desfavorable) del score SAMe-TT2R2.
Palabras clave:
FIBRILACIÓN ATRIAL
WARFARINA
ANTICOAGULANTES
VITAMINA K/uso terapéutico ]]>
VITAMINA K/antagonista & inhibidores
SAMe-TT2R2
Usefulness of the SAMe-TT2R2 score in the control of oral anticoagulation with warfarin in patients with non-valvular atrial fibrillation
Summary
Background: prevention of thromboembolic events is the main objective of oral anticoagulation treatment in non-valvular atrial fibrillation (AF) and either oral anticoagulation with vitamin K antagonists (VKA), like warfarin, or direct oral anticoagulants (ACOD) can be used. When warfarin is used, a time in therapeutic range (TTR) >65- 70% offers the best efficacy and safety. The SAMe-TT2R2 score has been developed as a tool to predict the response to VKA. A favorable SAMe-TT2R2 score (0-1 point) can identify patients that respond adequately to VKA and will have a better TTR, whilst a not favorable SAMe-TT2R2score (2 points) associates with poor TTR, needing additional therapies to optimize anticoagulation quality control or would be better candidates to direct oral anticoagulants (ACOD)
Objective: assess SAMe-TT2R2 score value in anticoagulation quality control of patients with non-valvular AF treated with warfarin.
Method: retrospective study of 115 ambulatory patients with non-valvular AF receiving oral anticoagulation treatment with warfarin, from June 1st 2012 to June 31st 2014. Analyzed variables were age, sex, left ventricle ejection fraction (LVEF), comorbidities, number of concomitant drugs, CHA2DS2-VASc and HAS-BLED scores. Rosendaal method was used to calculate the individual TTR. Student T test was used to compare mean values and X2 test for categorical variables. P < 0.05 was considered statistically significant.
Results: mean age was 71.0 ± 9.8 years, 52.2% were male, most frequent comorbidities were: hypertension 82.6%, ischemic heart disease 24.3%, diabetes mellitus 18.3%, cerebrovascular disease 10.4%, smoking 6.1% and former smoking 30.4%, concomitant use of 3 or more drugs 87%. Mean LVEF was 48.3 ± 12.5 %, CHA2DS2-VASc Score 3.6 ± 1.2 points and HAS-BLED Score 1.8 ± 0.9 points. Mean TTR was 54.9 ± 21.6% and only 37 patients (32.2%) had a TTR 65%. Mean SAMe-TT2R2 score was 1.8 ± 1.0 points, a warfarin-favorable score (0-1 point) was found in 45 patients (39.1%) and 70 patients (60.9%) had a non-favorable score (2 points). No significant mean TTR difference was found among SAMe-TT2R2 categories (53.0 ± 23.7% vs. 56.2 ± 20.2%, p=0.447). No association between a favorable SAMe-TT2R2 Score and a high TTR (65%) was found (33.3 vs. 31.4%, p=0.831). ]]>
Conclusion: no difference in anticoagulation quality control was found among favorable and non-favorable SAMe-TT2R2 score categories.
Key words:
ATRIAL FIBRILLATION
WARFARIN
ANTICOAGULANTS
VITAMIN K/therapeutic use
VITAMIN K/antagonists & inhibitors
SAMe-TT2R2
La fibrilación auricular (FA) es la arritmia más frecuente, con una prevalencia en la población general de 1%-2%, siendo en torno a 6% en personas 65 años y de 10% en personas 80 años(1-3). Datos similares han sido obtenidos en nuestro país(4).
El riesgo de ataque cerebrovascular (ACV) es cinco veces mayor en pacientes portadores de FA y uno de cada cinco ACV son atribuidos a esta arritmia(5). El riesgo anual de ACV en pacientes portadores de FA no valvular no es homogéneo y puede estimarse a través del score CHA2DS2VASc(6,7). La necesidad de terapia antitrombótica debe ser reevaluada periódicamente en base a la estimación del riesgo tromboembólico y riesgo de sangrado individual(8,9).
El tratamiento anticoagulante representa la estrategia que genera mayor reducción de los eventos tromboembólicos en pacientes con FA. En el metaanálisis de Hart y colaboradores(10), el tratamiento con warfarina estuvo asociado a una reducción del riesgo relativo de ACV de 64%, correspondiendo a una reducción de riesgo absoluto anual de 2,7% en todos los ACV. La reducción de riesgo fue similar tanto para prevención primaria como secundaria y en ACV invalidante y no invalidante. La prevención de eventos tromoboembólicos puede realizarse utilizando fármacos antivitamina K (AVK), como warfarina y anticoagulantes orales directos (ACOD)(11-16). El principal dilema que se plantea es la selección del tipo de anticoagulante oral en un escenario de igual accesibilidad para ambos.
La warfarina presenta conocidas limitaciones vinculadas a un estrecho rango terapéutico, múltiples interacciones farmacológicas y dietéticas así como también a una amplia variabilidad en su respuesta anticoagulante inter e intraindividuo(17). Para disminuir la incidencia de complicaciones trombóticas y hemorrágicas se debe mantener el INR (International Normalized Ratio) dentro del rango terapéutico indicado para cada patología(18-20). La calidad de la anticoagulación oral con warfarina ha sido evaluada en múltiples estudios a través del cálculo del porcentaje de tiempo en que el INR se encuentra dentro del rango terapéutico, expresado como la sigla en inglés TTR (Time in Therapeutic Range)(17,18,20-23) y calculado mediante interpolación lineal por el método de Rosendaal(24). La warfarina ofrece su mayor eficacia y seguridad cuando el TTR es mayor de 65%-70%(25-28), valores inferiores se han asociado a eventos adversos (ACV, hemorragias y mortalidad)(29,30). El score de riesgo hemorrágico HAS- BLED define a los pacientes con un INR lábil como aquellos con TTR <60%(31,32).
Los ACOD no requieren ajuste de dosis en base a un test de coagulación específico, pero la adherencia al tratamiento es crucial, ya que estos fármacos tienen una vida media menor que la warfarina. Sus limitaciones se vinculan a su mayor coste económico y eliminación renal en grado variable, desaconsejando su uso en presencia de enfermedad renal severa (clearance de creatinina [ClCr] <30 mL/min/1,73 m2) y con necesidad de ajuste de dosis en presencia de enfermedad renal moderada (ClCr 30-49 mL/min/1,73 m2)(8,9). Tienen metabolismo hepático, estando contraindicada su administración en pacientes con disfunción hepática severa (Child-Pugh C), requiriendo ajuste de dosis o discontinuación en presencia de terapia concomitante con inductores o inhibidores moderados y potentes del CYP3A4 (citocromo P450) y P-gp (glicoproteína de permeabilidad). Finalmente, el acceso limitado a idaracizumab (antídoto de dabigatrán) recientemente aprobado por la FDA (Food and Drug Administration)(33)y la EMA (European Medicines Agency)(34)y la ausencia de antídoto específico comercializado hasta la fecha para los inhibidores del factor Xa representan también una limitación(8,35-38).
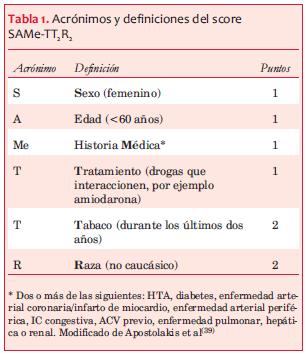
La selección del mejor anticoagulante oral para cada paciente particular es un desafío. El score SAMe-TT2R2(39) (sexo femenino, edad menor de 60 años, historia médica [dos o más comorbilidades], tratamiento [drogas que interaccionen, por ejemplo, amiodarona], tabaco [doble], raza no caucásica [doble]) (tabla 1) se ha desarrollado como herramienta para intentar predecir la respuesta al tratamiento anticoagulante con fármacos AVK. Aquellos pacientes con puntaje favorable a warfarina (0-1 punto) tendrían una buena respuesta al tratamiento y por lo tanto un TTR adecuado; mientras que un puntaje desfavorable ( 2 puntos) permitiría predecir un TTR inadecuado y en consecuencia mayores posibilidades de eventos adversos, identificando pacientes que requieren intervenciones adicionales para la optimización de la calidad de la anticoagulación con AVK o mejores candidatos a ACOD. Este score fue validado en diversos estudios(40-42) y su utilización se recomienda en una publicación reciente de la Sociedad Europea de Cardiología(26) y en un documento de consenso del NICE (National Institute for Health and Care Excellence)(28), sin embargo su reproducibilidad no ha sido consistente(43-46).
El objetivo de este estudio fue evaluar la utilidad del score SAMe-TT2R2 en el control de la anticoagulación oral con warfarina en pacientes portadores de FA no valvular.
]]>
 (39)
(39)
Material y método
Se realizó un estudio retrospectivo de una cohorte de 366 pacientes anticoagulados con warfarina que se asistían de forma ambulatoria en la Policlínica de Cardiología (283) y en la Unidad Multidisciplinaria de Insuficiencia Cardíaca (UMIC) (83) del Hospital de Clínicas entre el 1° de junio de 2012 y el 31 de junio de 2014.
Se incluyeron pacientes de ambos sexos mayores de 18 años que se encontraban bajo tratamiento anticoagulante por diagnóstico de FA no valvular de acuerdo con las guías de práctica clínica actuales(11).
Fueron excluidos aquellos que presentaban al menos uno de los siguientes criterios: (1) indicación adicional de anticoagulación por trombosis venosa profunda/tromboembolismo pulmonar (TVP/TEP) o trombo intracardíaco; (2) anticoagulación con ACOD; (3) inicio de anticoagulación menor a tres meses; (4) intervalo mayor a 60 días entre resultados de INR; (5) ausencia de cinco registros consecutivos.
Las variables analizadas fueron: edad, sexo, fracción de eyección del ventrículo izquierdo (FEVI), comorbilidades y fármacos concomitantes. Se evaluó el riesgo de ACV mediante el score CHA2DS2-VASc y el riesgo inicial de sangrado mediante el score HAS-BLED(31,32). Se evaluó la calidad de anticoagulación a través del cálculo individual del TTR mediante el método de Rosendaal (interpolación lineal)(24) y luego se determinó el resultado global expresado como la media de TTR. Se definió como adecuada calidad de anticoagulación un valor de TTR 65%.
]]> Resultados
En el período considerado se seleccionaron 115 pacientes que cumplían con los criterios propuestos, 45 (39,1%) provenientes de UMIC y 70 (60,9%) de la Policlínica de Cardiología. Las características de los pacientes se muestran en la tabla 2. La media de edad fue de 71,0±9,8 años y sexo masculino 52,2%. Las comorbilidades más frecuentes fueron: HTA 82,6%, cardiopatía isquémica 24,3% y diabetes mellitus 18,3%. El 87,0% de los pacientes consumía tres o más fármacos concomitantes. La media de FEVI fue 48,3±12,4%, el score CHA2DS2-VASc fue 3,6±1,2 puntos y el score HAS-BLED fue 1,8±0,9 puntos.
La calidad de la anticoagulación no fue óptima, siendo la media de TTR calculada 54,9±21,6% y solamente 37 pacientes (32,2%) tuvieron un TTR 65%. La media de días entre resultados de INR fue de 29,4±7,8 días. No se encontró asociación entre un TTR 65% y las variables estudiadas.
El score SAMe-TT2R2 tuvo una media de 1,8±1,0 puntos, 45 pacientes (39,1%) tuvieron un puntaje favorable a warfarina (0-1 punto) y 70 pacientes (60,9%) tuvieron un puntaje desfavorable (2 puntos). La relación entre la categoría del score SAMe-TT2R2 y las diferentes variables se muestra en la tabla 2.
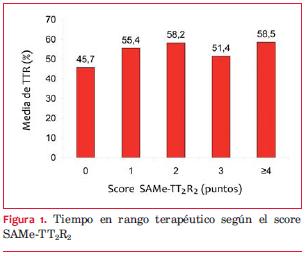
No hubo diferencia significativa en la media de TTR según la categoría del score SAMe-TT2R2 (53,0±23,7% vs 56,2±20,2%, p=0,447). En la categoría SAMe-TT2R2 favorable, solamente 15 pacientes (33,3%) tuvieron un TTR 65% (p=0,831). La puntuación del score SAMe-TT2R2 fue similar en los pacientes con TTR 65% y <65% (1,81±0,96 vs 1,81±1,1M; p=0,496). Tampoco se encontró diferencia entre la calidad de anticoagulación y un puntaje creciente en el score SAMe-TT2R2 (p=0,359) (figura 1).
]]>
Discusión
Considerando el score SAMe-TT2R2 como variable dicotómica (tabla 2) o como variable ordinal (figura 1) no se encontraron diferencias en la calidad de anticoagulación. La media de TTR no fue significativamente diferente entre los pacientes con un score SAMe-TT2R2 favorable a warfarina y uno desfavorable.
Los pacientes con score SAMe-TT2R2 favorable a warfarina tuvieron menor consumo de fármacos concomitantes y menor prevalencia de algunos factores de riesgo cardiovascular (HTA y tabaquismo) y de enfermedad cardiovascular definida (cardiopatía isquémica). Estos elementos pueden estar vinculados al hallazgo de una mayor FEVI y un menor score CHA2DS2-VASc. Sin embargo, ninguna de estas variables se asoció a un TTR 65%.
El score SAMe-TT2R2 (39), es propuesto como un score simple, conformado por variables clínicas y de fácil aplicación, siendo esa su principal fortaleza. Sin embargo, su utilidad es cuestionada por diversos estudios(43-45). En pacientes con FA no valvular e insuficiencia cardíaca, este score no permitió predecir el TTR tras un ingreso por descompensación(46).
Es importante considerar que el estudio inicial de Apostolakis y colaboradores(39) incluyó a todos los pacientes con indicación de anticoagulación oral del estudio AFFIRM(48), utilizándose en la fase de validación la misma cohorte de pacientes y existiendo 11% de pacientes con FA valvular(49). En dicho estudio se demostró que utilizando como punto de corte una media de TTR de 65%, el score SAMe-TT2R2 permitiría identificar a los pacientes con respuesta favorable a warfarina. Sin embargo, en nuestra cohorte, de los pacientes con puntaje favorable a warfarina (0-1 punto) solamente el 33% tuvo un TTR 65%. La incapacidad de detectar una diferencia se podría explicar debido a que la media de TTR calculada en nuestra población (54,9%) fue inferior al valor óptimo (>65%-70%). Este hallazgo de anticoagulación subóptima coincide con el obtenido en el registro GARFIELD-AF que muestra una media de TTR de 55,8% en pacientes tratados con warfarina provenientes de la “vida real”(50). Una pregunta a responder es si este score tiene utilidad en poblaciones con anticoagulación subóptima. La publicación de Abumuaileq y colaboradores(42), que evalúa la calidad de anticoagulación calculando el porcentaje de INR dentro del rango terapéutico (PINRR), no utilizando el TTR, pone en evidencia que el rendimiento del score varía de acuerdo al punto de corte de PINRR utilizado. En pacientes con PINRR 70%, el 82,1% tenía un puntaje del score SAMETTR favorable (0-1 punto) y este porcentaje se reducía de forma significativa al considerar un punto de corte de PINRR <70% (70,9% para un PINRR 65% y 70,7% para un PINRR 60%, p<0,001).
Existe además cierta disparidad en la literatura con respecto al punto de corte utilizado para definir las diferentes categorías. En la publicación de Apostolakis y colaboradores(39) se considera que los pacientes con un puntaje de 0-1 tendrían una respuesta favorable al tratamiento con warfarina, mientras que aquellos pacientes con 2 puntos tendrían una respuesta desfavorable. Numerosos estudios utilizan este criterio(42-46). Por otro lado, algunas publicaciones consideran un resultado de 0-2 puntos como favorable a warfarina (0-1 punto: riesgo bajo y 2 puntos: riesgo intermedio) mientras que un puntaje mayor de 2 puntos sería desfavorable, con alto riesgo de pobre TTR(40,41,51,52). Es necesaria información adicional para definir adecuadamente las diferentes categorías del score y evaluar su validez en diferentes poblaciones.
Las guías de práctica clínica para la prevención de eventos tromboembólicos en pacientes con FA no valvular avalan la utilización de fármacos AVK y nuevos anticoagulantes orales (NACO)(8,9). Sin embargo, en el estudio ROCKET-AF(14) (rivaroxabán), el grupo control tratado con AVK tenía una media de TTR de 55%, en el estudio RE-LY(15) (dabigatrán) 64% y en el estudio ARISTOTLE(16) (apixabán) fue de 62%, reflejando niveles subóptimos de anticoagulación incluso en poblaciones “controladas”. Debemos considerar que si bien los ACOD ofrecen ciertas ventajas, su superioridad en poblaciones con TTR mayor a 70% no ha sido evaluada.
Implicancias
Este estudio nos permite conocer la calidad de la anticoagulación y las características de los pacientes con FA no valvular, siendo el TTR el parámetro recomendado para evaluarla(21) y con criterios de inclusión que aseguran que el TTR calculado realmente refleje la calidad de la anticoagulación. La media de TTR obtenida es inferior al valor óptimo, siendo similar al resultado obtenido en un estudio previo realizado en Uruguay (56,6%)(53). Un incremento absoluto en la media de TTR mayor a 5% representaría una diferencia significativa y clínicamente relevante(54). Debemos, por lo tanto, estimular el desarrollo de nuevas estrategias educativas y de control de anticoagulación para optimizar el nivel de la misma.
Limitaciones
Están vinculadas principalmente a su pequeño tamaño y ser unicéntrico, siendo sus resultados, por ende, no generalizables. Desconocemos cuál es la calidad de la anticoagulación de los pacientes con FA no valvular de otros centros de tercer nivel así como en policlínicas de atención primaria. Se han excluido aquellos pacientes con FA no valvular que no reciben tratamiento anticoagulante oral o que reciben ACOD, lo cual puede representar un sesgo de selección. Dichos pacientes deberían ser considerados en estudios futuros.
Al ser retrospectivo, no se ha evaluado la incidencia de eventos tromboembólicos, hemorrágicos y muerte cardiovascular. El registro de dichos eventos permitiría evaluar si existe asociación con el puntaje de los scores CHA2DS2-VASc y HAS-BLED, media de TTR obtenido y la categoría del score SAMe-TT2R2. En un estudio reciente de Lip y colaboradores(51) un score SAMe-TT2R2 >2 se ha visto asociado con un riesgo incrementado de INR lábil (p=0,004), ACV/tromboembolismo (p=0,007), sangrado grave (p<0,0001) y muerte (p=0,002) en el seguimiento.
Conclusión
En la población estudiada no hubo diferencias en la calidad de la anticoagulación oral con warfarina entre las categorías (favorable y desfavorable) del score SAMe-TT2R2.
Bibliografía
1. Stewart S, Hart CL, Hole DJ, McMurray JJ. Population prevalence, incidence, and predictors of atrial fibrillation in the Renfrew/Paisley study. Heart. 2001;86(5):516-521.
2. Go AS, Hylek EM, Phillips KA, Chang Y, Henault LE, Selby JV, et al. Prevalence of diagnosed atrial fibrillation in adults. JAMA 2001; 285(18): 2370-2375.
3. Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, et al. Executive summary: heart disease and stroke statistics—2010 update: a report from the American Heart Association. Circulation. 2010;121(7):948-54.
4. Sandoya E, Aguilar MP, Vázquez H. Prevalencia de la fibrilación auricular en la población adulta de Montevideo. Rev Urug Cardiol. 2014;29(2):187-91.
5. Kannel W, Wolf P, Benjamin E, Levy D. Prevalence, incidence, prognosis, and predisposing conditions for atrial fibrillation: population-based estimates. Am J Cardiol 1998;82(7):2N-9N.
6. Lip GYH, Nieuwlaat R, Pisters R, Lane DA, Crijns HJGM. Refining clinical risk stratification for predicting stroke and thromboembolism in atrial fibrillation using a novel risk factor-based approach: the euro heart survey on atrial fibrillation. Chest. 2010;137(2):263-72.
7. Lip GYH, Frison L, Halperin JL, Lane DA. Identifying patients at high risk for stroke despite anticoagulation: a comparison of contemporary stroke risk stratification schemes in an anticoagulated atrial fibrillation cohort. Stroke 2010;41(12):2731-8.
8. Camm AJ, Lip GY, De Caterina R, Savelieva I, Atar D, Hohnloser SH, et al. 2012 focused update of the ESC Guidelines for the management of atrial fibrillation. Eur Heart J 2012;33(21):2719-47.
9. January CT, Wann LS, Alpert JS, Calkins H, Cigarroa JE, Cleveland JC Jr, et al. 2014 AHA/ACC/HRS Guideline for the management of patients with atrial fibrillation. J Am Coll Cardiol. 2014;64(21):e1-e76.
10. Hart RG. Meta-analysis: antithrombotic therapy to prevent stroke in patients who have nonvalvular atrial fibrillation. Ann Intern Med. 2007; 146(12): 857.
11. Ahrens I, Lip G, Peter K. New oral anticoagulant drugs in cardiovascular disease. Thromb Haemost. 2010; 104(1):49-60. Disponible en: http:file:///D:/Mis%20documentos/Downloads/th_2010-104- 1_13189.pdf (Consultado 30 Abril 2015).
12. Connolly SJ, Eikelboom J, Joyner C, Diener HC, Hart R, Golitsyn S, et al. Apixaban in patients with atrial fibrillation. N Engl J Med 2011; 364(9):806-17.
13. Connolly SJ, Ezekowitz MD, Yusuf S, Eikelboom J, Oldgren J, Parekh A, et al. Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 2009;361(12):1139-51.
14. Patel MR, Mahaffey KW, Garg J, Pan G, Singer DE, Hacke W, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 2011;365(10):883-91.
15. Flaker G, Ezekowitz M, Yusuf S, Wallentin L, Noack H, Brueckmann M, et al. Efficacy and safety of dabigatran compared to warfarin in patients with paroxysmal, persistent, and permanent atrial fibrillation: results from the RE-LY (Randomized evaluation of long-term anticoagulation therapy) study. J Am Coll Cardiol 2012;59(9):854-5.
16. Granger CB, Alexander JH, McMurray JJ V, Lopes RD, Hylek EM, Hanna M, et al. Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 2011;365(11):981-92.
17. Veeger NJGM, Piersma-Wichers M, Tijssen JGP, Hillege HL, Van Der Meer J. Individual time within target range in patients treated with vitamin K antagonists: main determinant of quality of anticoagulation and predictor of clinical outcome. A retrospective study of 2300 consecutive patients with venous thromboembolism. Br J Haematol 2005; 128(4):513-9.
18. Wan Y, Heneghan C, Perera R, Roberts N, Hollowell J, Glasziou P, et al. Anticoagulation control and prediction of adverse events in patients with atrial fibrillation: a systematic review. Circ Cardiovasc Qual Outcomes 2008;1(2):84-91.
19. Ansell J, Hirsh J, Hylek E, Jacobson A, Crowther M, Palareti G. Pharmacology and management of the vitamin K antagonists: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest 2008;133(6 Suppl): 160S-198S.
20. Phillips KW, Ansell J. Outpatient management of oral vitamin K antagonist therapy: defining and measuring high-quality management. Expert Rev Cardiovasc Ther 2008;6(1):57-70.
21. Hutten BA, Prins MH, Redekop WK, Tijssen JG, Heisterkamp SH, Büller HR. Comparison of three methods to assess therapeutic quality control of treatment with vitamin K antagonists. Thromb Haemost 1999;82(4):1260-3. Disponible en: http://www.ncbi.nlm.nih.gov/pubmed/10544910. (Consultado 30 Abril 2015).
22. Erkens PMG, ten Cate H, Büller HR, Prins MH. Benchmark for time in therapeutic range in venous thromboembolism: a systematic review and meta- analysis. PLoS One. 2012;7(9):e42269.
23. Locadia M, Van Geest-Daalderop JHH, Sprangers MAG, Hutten BA, Prins MH. The relationship between adherence and quality of treatment with vitamin K antagonists. J Thromb Haemost 2004; 2(2): 362-3.
24. Rosendaal FR, Cannegieter SC, van der Meer FJ, Briët E. A method to determine the optimal intensity of oral anticoagulant therapy. Thromb Haemost. 1993;69(3):236-9.
25. Connolly SJ, Pogue J, Eikelboom J, Flaker G,Commerford P, Franzosi MG, et al. Benefit of oral anticoagulant over antiplatelet therapy in atrial fibrillation depends on the quality of international normalized ratio control achieved by centers and countries as measured by time in therapeutic range. Circulation. 2008;118(20):2029-37.
26. De Caterina R, Husted S, Wallentin L, Andreotti F, Arnesen H, Bachmann F, et al. Vitamin K antagonists in heart disease: current status and perspectives (Section III). Position paper of the ESC Working Group on Thrombosis—Task Force on Anticoagulants in Heart Disease. Thromb Haemost. 2013;110(6):1087-107.
27. De Caterina R, Husted S, Wallentin L, Andreotti F, Arnesen H, Bachmann F, et al. General mechanisms of coagulation and targets of anticoagulants (Section I). Position paper of the ESC Working Group on Thrombosis—Task Force on Anticoagulants in Heart Disease. Thromb Haemost 2013; 109(4):569-79.
28. National Institute for Health and Care Excellence. Atrial fibrillation: the management of atrial fibrillation (clinical guideline 180). 2014. Disponible en: https://www.nice.org.uk/guidance/cg180.
29. Gallagher AM, Setakis E, Plumb JM, Clemens A, van Staa T-P. Risks of stroke and mortality associated with suboptimal anticoagulation in atrial fibrillation patients. Thromb Haemost. 2011; 106(5):968-77.
30. Morgan CL, McEwan P, Tukiendorf A, Robinson PA, Clemens A, Plumb JM. Warfarin treatment in patients with atrial fibrillation: observing outcomes associated with varying levels of INR control. Thromb Res 2009;124(1):37-41.
31. Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJGM, Lip GYH. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest 2010;138(5):1093-100.
32. Lip GYH, Frison L, Halperin JL, Lane DA. Comparative validation of a novel risk score for predicting bleeding risk in anticoagulated patients with atrial fibrillation: the HAS-BLED (Hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile INR, elderly, drug. J Am Coll Cardiol 2011;57(2):173-180.
33. FDA. FDA approves Praxbind, the first reversal agent for the anticoagulant Pradaxa. News Release, . Oct 16, 2015. Silver Spring, MD : FDA; 2015. [updated 2015 Oct. 19]. Disponible en: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm467300.htm.
34. European Medicines Agency. Praxbind: idarucizumab. London: EMA; c1995-2015. [updated 2015 Dec. 1]. Disponible en: http://www.ema.europa.eu/ema/index.jsp?curl=pages/medicines/human/medicines/003986/human_med_001938.jsp&mid=WC0b01ac058001d124.
35. Huisman M V, Lip GYH, Diener H-C, Brueckmann M, van Ryn J, Clemens A. Dabigatran etexilate for stroke prevention in patients with atrial fibrillation: resolving uncertainties in routine practice. Thromb Haemost. 2012;107(5):838-47.
36. Tripodi A. Measuring the anticoagulant effect of direct factor Xa inhibitors. Is the anti-Xa assay preferable to the prothrombin time test? Thromb Haemost 2011;105(4):735-6.
37. Barrett YC, Wang Z, Frost C, Shenker A. Clinical laboratory measurement of direct factor Xa inhibitors: anti-Xa assay is preferable to prothrombin time assay. Thromb Haemost 2010;104(6):1263-71.
38. Pengo V, Crippa L, Falanga A, Finazzi G, Marongiu F, Palareti G, et al. Questions and answers on the use of dabigatran and perspectives on the use of other new oral anticoagulants in patients with atrial fibrillation. A consensus document of the Italian Federation of Thrombosis Centers (FCSA). Thromb Haemost 2011;106(5):868-76.
39. Apostolakis S, Sullivan RM, Olshansky B, Lip GYH. Factors affecting quality of anticoagulation control among patients with atrial fibrillation on warfarin: the SAMe-TTR score. Chest. 2013;144(5): 1555-63.
40. Poli D, Antonucci E, Testa S, Lip GYH. A prospective validation of the SAME-TT2R 2 score: how to identify atrial fibrillation patients who will have good anticoagulation control on warfarin. Intern Emerg Med. 2014;9(4):443-7.
41. Gallego P, Roldán V, Marin F, Gálvez J, Valdés M, Vicente V, et al. SAME-TT2R2 score, time in therapeutic range and outcomes in anticoagulated patients with atrial fibrillation. Am J Med 2014; 127(11):1083-8.
42. Abumuaileq RR-Y, Abu-Assi E, Raposeiras-Roubin S, López-López A, Redondo-Diéguez A, Álvarez-Iglesias D, et al. Evaluation of SAMe- TT2R2 risk score for predicting the quality of anticoagulation control in a real-world cohort of patients with non-valvular atrial fibrillation on vitamin-K antagonists. Europace 2015;17(5):711-7.
43. Skov J, Bladbjerg E-M, Bor MV, Gram J. SAMe-TT2R2 does not predict time in therapeutic range of the international normalized ratio in patients attending a high-quality anticoagulation clinic. Chest 2014;145(1):187-8.
44. Park YK, Lee MJ, Kim JH, Kim SJ, Kim JS, Lee SY, et al. Lack of Association of Clinical Factors (SAMe- TT2R2) with CYP2C9/VKORC1 Genotype and Anticoagulation Control Quality. J Stroke. 2015;17(2):192-8.
45. Tigera U T, Fernández M, Pérez R, Hermosa V, Neila Calvo S, Sánchez González A, et al. Utilidad del score SAME-TT2R2 en el control de la anticoagulación con acenocumarol en una muestra de pacientes con fibrilación auricular no valvular en un hospital universitario de tercer nivel. Rev Clin Esp 2014:214 (Espec Congr):785.
46. Andreu-Cayuelas J, Puche C, Caro-Martínez C, Flores-Blanco M, Manzano-Fernández S. La puntuación SAMe-TT2R2 no predice el tiempo en rango terapéutico tras un ingreso por insuficiencia cardiaca aguda en pacientes con fibrilación auricular. Rev Esp Cardiol. 2016; 69(4): 453-4. Avance On - Line. Disponible en: http://file:///D:/Mis%20documentos/Downloads/S0300893216000312_ S200_es.pdf. (Consultado 3 marzo 2016).
47. Camm AJ, Kirchhof P, Lip GYH, Schotten U, Savelieva I, Ernst S, et al. Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J. 2010; 31(19):2369-429.
48. Wyse DG, Waldo AL, DiMarco JP, Domanski MJ, Rosenberg Y, Schron EB, et al. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med. 2002;347 (23):1825-33.
49. Zhang H, Yang Y, Zhu J. The SAMe-TT2R2 score: far from clinical application. Chest. 2014;145(2): 418-9.
50. Hass S. Time in therapeutic range and frequency in range with 1-year outcomes in cohorts 1-3 of GARFIELD-AF. London: Thrombosis research Institute London; 2015 [consulta 21 Set. 2016]. Disponible en: http://congress365.escardio.org/Presentation/120023#.V-MQBTXSPCZ
51. Lip GYH, Haguenoer K, Saint-Etienne C, Fauchier L. Relationship of the SAMe-TT2R2 score to poor-quality anticoagulation, stroke, clinically relevant bleeding, and mortality in patients with atrial fibrillation. Chest. 2014;146(3):719-726.
52. Proietti M, Lip GYH. Simple decision-making between a vitamin K antagonist and a non-vitamin K antagonist oral anticoagulant: using the SAMe-TT 2 R 2 score. Eur Heart J - Cardiovasc Pharmacother. 2015;1(3):150-152.
53. Gómez A, Peixoto S, Azcúnaga M, Gama Alejandra, Lluberas N, Álvarez P. Calidad de la anticoagulación oral con warfarina en una policlínica de cardiología: porcentaje de tiempo en rango terapéutico. Rev Urug Cardiol. 2014;29:311-6.
54. Samsa GP, Matchar DB. Relationship between test frequency and outcomes of anticoagulation: a literature review and commentary with implications for the design of randomized trials of patient self-management. J Thromb Thrombolysis 2000;9(3): 283-92.