 ]]>
]]>
CASO CLÍNICO
Arch Pediatr Urug 2008; 79(4)
Cierre percutáneo de la comunicación interauricular con aneurisma del septum.
A propósito de un caso clínico
Juan Carlos Gambetta 1, Pedro Chiesa 2, Javier Giudice 3, Jorge Morales 4,
Sucy Dutra 1, Daniela Denegri 5, Carlos Peluffo 6, Pedro Duhagon 7
1. Pediatra. Cardiólogo Pediatra.
2. Cardiólogo Pediatra. Hemodinamista. ]]>
3. Pediatra. Intensivista Pediátrico.
4. Cardiólogo Pediatra. Ecocardiografista.
5. Licenciada Neumocardióloga.
6. Cardiólogo Pediatra. Director Técnico del ICI.
7. Cardiólogo Pediatra. Ex Prof. Agdo. Pediatría.
Servicio de Cardiología Pediátrica. Hospital Pereira Rossell. Instituto de Cardiología Infantil (ICI). MUCAM. Montevideo Uruguay.
Fecha recibido: 15 de setiembre de 2008
Fecha aprobado: 10 de diciembre de 2008
El cierre de los defectos del septum interauricular (CIA) tipo ostium secundum mediante el uso de dispositivos de implante por vía percutánea se realiza actualmente con seguridad y eficacia. En nuestro medio el dispositivo autorizado es el Amplatzer ASD (AGA Minneapolis EE.UU.). El éxito en su uso está dado por la selección adecuada de los pacientes. Se presenta una adolescente de 14 años portadora de una CIA tipo ostium secundum con aneurisma del septum interauricular amplio; cumpliendo con los criterios de indicación para el cierre con dispositivo implantado por vía percutánea. El aneurisma del septum no contraindica el cierre con dispositivo, representando un reto en la maniobra de colocación y liberación del mismo. El cierre se realizó con éxito sin complicaciones y dando de alta a la paciente a las 18 horas del procedimiento sin shunt residual.
Palabras clave:
DEFECTOS DEL TABIQUE INTERATRIAL
CATETERISMO CARDÍACO
Summary
The closure of interauricular septal defects type ostium secundum through percutaneous implantation devices is nowadays done in a secure and efficient manner. The Amplatzer ASD (AGA Minneapolis USA) is the authorized device used in our media. The success in its use is determined by the appropriate choice of the patients. We present the case of a 14 year old adolescent (female) with ostium secundum and a large interauricular septum aneurysm, which fulfilled the selection criteria and showed no contraindications for the closure with percutaneous device implantation. The septum aneurysm is not a contraindication for closure with a device; however it represents a challenge in its manoeuvering and placement. The closure was performed successfully and without further complications. The patient was discharged 18 hours after the procedure, without residual shunt.
Key words:
HEART SEPTAL DEFECTS, ATRIAL ]]>
HEART CATHETERIZATION
Introducción
La experiencia en el cierre de la CIA tipo ostium secundum mediante el uso de dispositivos implantados por vía percutánea está bien establecida (1-3,4) con resultados comparables a la cirugía (5-7). El dispositivo más ampliamente usado en la actualidad es el Amplatzer ASD por ser un sistema seguro y eficaz en manos expertas; se ha demostrado una mortalidad nula y una morbilidad menor en comparación con el tratamiento quirúrgico (8-10). Un reto especial del cierre percutáneo de los defectos interauriculares constituyen los grandes defectos, los orificios múltiples y los aneurismas (11,12). El aneurisma del septo interauricular en la CIA tipo ostium secundum o foramen oval permeable tiene un elevado riesgo de embolia paradojal y recurrencia en el stroke neurológico (4,4% anual) (13). Las anomalías asociadas que requieren sanción quirúrgica y la hipertensión arterial pulmonar con resistencias pulmonares elevadas contraindican el procedimiento (14,15).
Objetivo
Presentar un caso clínico de cierre percutáneo de una comunicación interauricular amplia tipo ostium secundum con aneurisma del septum.
Caso clínico
Paciente de 14 años, sexo femenino, raza blanca, procedente de Florida. Buen crecimiento y desarrollo. Sin antecedentes personales patológicos a destacar. Consulta por dolor torácico. Al examen físico se destaca a nivel cardiovascular un ritmo regular, desdoblamiento fijo del segundo ruido en el borde esternal superior izquierdo, soplo sistólico grado 2/6 sin irradiación en igual topografía. Resto del examen físico sin particularidades. El ECG mostró un ritmo sinusal con bloqueo incompleto de rama derecha del haz de His. En la radiografía de tórax se observó cardiomegalia a expensas de cavidades derechas con hiperflujo pulmonar. El ecocardiograma Doppler color mostró una CIA tipo ostium secundum de 8 mm con aneurisma del septum interauricular amplio, dilatación de aurícula y ventrículo derechos (27 mm, normal: 8 a 17 mm). La distancia de los bordes de la CIA a vena pulmonar superior derecha, seno coronario, vena cava inferior y válvula mitral fueron mayores a 7 mm. Prolapso de válvula mitral leve con mínima insuficiencia.
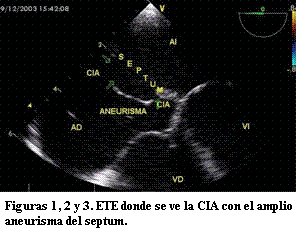
Se efectuó cateterismo de coordinación bajo anestesia general, con monitorización de ECG basal, presión arterial sistémica por método no invasivo y saturación arterial por oxímetro de pulso. Se colocó sonda para ecografía transesofágica (ETE) evaluando los hallazgos del ecocardiograma transtorácico (ETT): CIA tipo ostium secundum con aneurisma del septum interauricular, tamaño y consistencia de los bordes del defecto, confirmando que son adecuados para el cierre con dispositivo y se descartaron malformaciones asociadas (figuras 1, 2 y 3). Mediante acceso por vena femoral derecha se obtienen los registros hemodinámicos basales, medida de la presión arterial pulmonar, cálculo de la magnitud del cortocircuito mediante el método de Fick calculando la relación flujo pulmonar/flujo sistémico (QP/QS) y cálculo de las resistencias pulmonares. Se realizan angiografías en la arteria pulmonar y en la vena pulmonar superior derecha para valorar la topografía y el tamaño del defecto (figura 4).
A continuación se coloca balón medidor a través de la CIA procediendo a realizar su insuflación controlado por ETE (figura 5), momento en el cual se hace la medida del defecto estirado que fue de 16 mm (figura 6).
Una vez determinada la indicación del cierre con dispositivo se procede a elegir el tipo y tamaño del mismo. En este caso se utilizó un dispositivo Amplatzer ASD por ser el que cumple con las mejores cualidades según la literatura internacional (9) y es el autorizado en nuestro medio. El oclusor septal Amplatzer ASD es un dispositivo de doble disco que se autoexpande y está hecho de malla de nitinol (aleación de níquel y titanio) rellena de tela de poliéster. Los dos discos están unidos entre sí por una pieza central corta de conexión. En la figura 7 se muestra un esquema del dispositivo.
El tamaño elegido fue de 16 mm en base al diámetro del defecto; también se considera el tamaño del disco atrial izquierdo en relación a la medida del diámentro del septum. Para un dispositivo de 16 mm el disco auricular izquierdo es 14 mm mayor, totalizando 30 mm para un diámetro septal interauricular de 31 mm en este caso.Se implanta a través de una vaina de Mullins posicionada en aurícula izquierda teniendo sumo cuidado en evitar la introducción de burbujas de aire hacia la circulación sistémica, el dispositivo es desplegado a nivel del defecto septal y aun sujeto al cable de liberación se comprueba la correcta posición del mismo y la ausencia de compromiso de estructuras vecinas por ETE. Luego se lo libera con control radioscópico y por ETE, comprobando la presencia de la membrana del aneurisma dentro de los discos del dispositivo y la existencia de un insignificante shunt residual a través del mismo (normal en las primeras horas luego de la colocación) (figura 8).
La paciente permaneció internada por 18 horas, se efectuó antiagregación plaquetaria con AAS 100 mg/día por vía oral que se mantendrá por 6 meses, siendo dada de alta en buenas condiciones hemodinámicas con radiografía de tórax que mostró el dispositivo bien posicionado y el ETT reveló la ausencia total del shunt residual. Se indicó restricción de la actividad física por un mes.
Discusión
El tratamiento clásico de la CIA tipo ostium secundum ha sido el cierre quirúrgico bajo circulación extracorpórea. Con el objetivo de establecer un tratamiento menos agresivo, con menor mortalidad, menor costo y menor tiempo de internación, se han desarrollado diferentes dispositivos para el cierre percutáneo. El dispositivo más usado es el Amplatzer ASD por sus condiciones de flexibilidad, elasticidad, memoria (posibilidad de reintroducirlo dentro de la vaina cuando el despliegue es inadecuado y reposicionarlo nuevamente), y disponibilidad de una amplia variedad de tamaños (16).
El uso de la ETE juega un importante rol, permitiendo cuantificar el tamaño del defecto, la valoración de los bordes, la relación con estructuras vecinas (vena pulmonar superior derecha, válvula mitral, seno coronario, aorta) medida con balón, despliegue del dispositivo y su liberación (17).
El caso clínico presentado tiene indicación para el cierre de la CIA por el importante cortocircuito de izquierda a derecha con dilatación de cavidades derechas y relación QP/QS mayor a 1,5:1 y por tratarse de una CIA de tipo ostium secundum con bordes apropiados. En las tablas 1 y 2 se exponen las indicaciones y contraindicaciones de cierre de la CIA con dispositivo (1,14,15,18-20).



Al efectuar el cierre de una CIA con aneurisma del septum se deben tener más precauciones, por la eventualidad de desgarros del septum aneurismático con embolización, ya sea en las maniobras de cateterismo o durante el insuflado del balón medidor (por lo que esta maniobra debe ser realizada con sumo cuidado y con baja presión); también riesgo de migración del dispositivo sea en forma inmediata o alejada por una inadecuada fijación.
La técnica de cierre con dispositivo implantado por vía percutánea se destaca por su menor costo, menor tiempo de internación, menor agresión psicofísica y por presentar una menor morbilidad que el cierre por cirugía, a tener en cuenta en el momento de la toma de decisiones. En la tabla 3 se exponen las posibles complicaciones del cierre de CIA con dispositivo.
Conclusiones
Se presentó el caso clínico de una adolescente de 14 años portadora de una CIA de tipo ostium secundum con aneurisma del septum interauricular, con presión arterial pulmonar normal, dilatación y sobrecarga de volumen de cavidades derechas con indicación de oclusión del defecto con dispositivo implantado por vía percutánea. Se utilizó un dispositivo oclusor Amplatzer ASD de 16 mm, procedimiento fue eficaz, sin complicaciones y sin shunt residual. El cierre percutáneo con dispositivo de los defectos del septum auricular constituye una herramienta segura y eficaz, aun en presencia de aneurisma del septum, orificios múltiples o los grandes defectos, constituyendo éstos un reto especial para el cierre percutáneo.
Referencias bibliográficas
]]>1. Rigby M. The era of transcatheter closure of atrial septal defects. Heart 1999; 81: 227-8.
2. King TD, Mills NL. Secundum atrial septal defects: non operative closure during cardiac catheterization. J Am Med Ass 1976; 235: 2506-9.
3. Lock JE, Rome JJ, Davis R, Van Praagh S, Perry SB, Van Praagh R, et al. Transcatheter closure of atrial septal defects: experimental studies. Circulation 1989; 79: 1091-9.
4. Chiesa P, Giúdice J, Morales J, Gambetta JC, Peluffo C, Duhagón P. Diez años de experiencia en el cierre percutáneo de la comunicación interauricular y del ductus arterioso persistente. Rev. Urug Cardiol 2008; 23 (1): 84-9.
5. Durongpisitkul K, Soongswang J, Laohaprasitiporn D, Nana A, Sriyoschati S, Ponvilawan S, et al. Comparison of Atrial Septal Defect Closure Using Amplatzer Septal Occluder with Surgery. Pediatr Cardiol 2002; 23: 36-40.
]]>6. Kim JJ, Hijazi ZM. Clinical outcumes and costs of Amplatzer transcatheter closure as compared with surgical closure of ostium secundum atrial septal defects. Med Sci Monit 2002; 8(12): 787-91.
7. Berger F, Vogel M, Alexi-Meskishivili V, Lange PE. Comparision of results and Complication of surgical and Amplatzer device closure of atrial septal defects. J Thorac Cardiovasc Surg. 1999; 118(4): 674-80.
8. Du ZD, Hijazi Z, Kleinman C, Silverman N, Larnz K. For de Amplatzer investigators. Comparison between transcatheter and surgical closure of secundum atrial defect in children and adults. J Am Coll Cardiol 2002; 39; 1836-44.
9. Masura J, Gavora P, Formanek A, Hijaz ZM. Transcatheter closure of secundum atrial septal defects using the new self-centering Amplatzer septal ocluder: initial human experience. Cathet Cardiovasc Diagn 1997; 42: 388-93.
10. Cham KC, Godman MJ, Walsh K, Wilson N, Redington A, Gibbs JL. Transcatheter closure of atrial septal defect and interatrial communications with a new self expanding nitinol double disc device. (Amplatzer septal ocluder): multicentre UK experience. Heart 1999; 82: 300-6.
]]>11. Suarez de Lezo J, Medina A, Pan M, Romero M, Segura J, Pavlovik D, et al. Transcatheter occlusion of complex atrial septal defect. Cathet Cardiovasc Intervent 2000; 51: 33-41.
12. Medina A, Suarez de Lezo J, Delgado A, Caballero E, Segura J, Romero M. Combined percutaneus atrial septal defects occlusion and pulmonary baloon valvuloplasty in adult patients. Tex Heart Inst J 2000; 27: 216-7.
13. Mas JL, Zuber M. Recurrent cerebrovascular events in patients with patent foramen ovale, atrial septal aneurysm, or both and cryptogenic stroke or transient ischemic attack. French Study Group on Patent Foramen Ovale and Atrial Septal Aneurysm. Am Heart J 1995; 130 (5); 1083-8.
14. Rao PS. Transcatheter closure of atrial septal defect: are we there yet? J Am Coll Cardiol 1998; 31: 1117-9.
15. Agarwal SK, Ghosh PK, Mittal PK. Failure of devices used for closure of atrial septal defects: Mechanisms and management. J Thorac Cardiovasc Surg 1996; 112: 21-6.
]]>16. Fernández A, del Cerro Marin MJ, Rubio D, Castro M, Granados F. Percutaneus closure of atrial septal defect whith the Amplatzer device: initial result and mid-term folow-up. Rev Esp Cardiol 2001; 54: 1190-6.
17. Figueroa MI, Blaguru D, McClure C, Kline CH, Radtke WAK, Shirali GS. Experience with use of multiplane transesophageal echocardiography to guide closure of atrial septal defects using the Amplatzer device. Pediatr Cardiol 2002; 23: 430-6.
18. Ries MW, Kampmann C, Rupprecht HJ, Hintereder G, Hafner G, Meyer J. Nickel release after implantation of the Amplatzer occluder. Am Heart J 2003; 145(4): 737-41.
19. Verma PK, Thingnam SK, Sharma A, Taneja JS, Varma JS, Grover A. Delayed embolization of Amplatzer septal occluder device: an unknown entity- a case report. Angiology 2003; 54(1): 115-8.
20. Hessling G, Hyca S, Hessling G, Hyca S, Brockmeier K, Ulmer HE. Cardiac dysrhythmias in pediatric patients before and 1 year after transcatheter closure of atrial septal defects using the Amplatzer septal occluder. Pediatr Cardiol 2003; 24(3): 259-62.
]]> Correspondencia: Dr. Juan Carlos Gambetta
Correo electrónico:juancarlosgambetta@gmail.com