Services on Demand
Journal
Article
Related links
Share
Agrociencia (Uruguay)
Print version ISSN 1510-0839On-line version ISSN 2301-1548
Agrociencia Uruguay vol.14 no.2 Montevideo Dec. 2010
Fusarium Species Recovered from Wheat and Barley Grains in Uruguay, Pathogenicity and Deoxynivalenol Content
Pereyra, Silvia1; Dill-Macky, Ruth2
1 Instituto Nacional de Investigación Agropecuaria, INIA La Estanzuela, 70000, Colonia, Uruguay. E-mail:spereyra@inia.org.uy
2 Department of Plant Pathology, University of Minnesota, St. Paul, MN 55108, USA.
Recibido: 18/8/09 Aceptado: 27/8/10
Summary
Grain samples from five wheat and five barley cultivars from different locations and planting dates were examined for the presence of Fusarium species. Fusarium graminearum was the primary species associated with FHB (Fusarium head blight). It comprised 76 % and 60 % of all Fusarium species isolated from wheat grains in 2001 and 2002, respectively. Fusarium graminearum represented 65 % and 56 % of all Fusarium species isolated from barley grains in 2001 and 2002, respectively. The frequencies with which Fusarium species other than F. graminearum were recovered varied depending on both environment and host cultivar. In general, F. avenaceum, F. culmorum and F. poae were the following most common species isolated from wheat grains, whereas F. equiseti, F. acuminatum, and F. trincictum were less frequently isolated. Fusarium poae and F. equiseti were the most common species isolated from barley grains after F. graminearum. Other Fusarium species recovered in barley grains included F. avenaceum, F. sambucinum, F. trincictum, F. semitectum, and F. chlamydosporum. All species were pathogenic on wheat and barley in inoculation tests in the greenhouse, except F. semitectum on wheat. Greater FHB severity and incidence on wheat and barley spikes were obtained with the F. graminearum isolates, followed by F. avenaceum and F. poae. Cultivars previously characterized as moderately resistant to moderately susceptible showed the lowest FHB incidences, severities, percentages of Fusarium-infested grains, and grains infested with F. graminearum in this field study. Data from this study raise the concern of the possible presence of mycotoxins other than DON (deoxynivalenol) in wheat and barley grains.
Key words: Fusarium head blight, Fusarium graminearum, Triticum aestivum, Hordeum vulgare, deoxynivalenol
Resumen
Especies de Fusarium recuperadas de granos de trigo y cebada en Uruguay, patogenicidad y contenido de deoxinivalenol
Se examinaron granos provenientes de cinco cultivares de trigo y cinco de cebada, de distintas localidades y fechas de siembra, por la presencia de especies de Fusarium. Fusarium graminearum fue la especie predominantemente asociada a FE (Fusariosis de la espiga) tanto en trigo como en cebada. La misma constituyó el 76 % y 60 % de todas las especies de Fusarium aisladas de los granos de trigo en 2001 y 2002, respectivamente y representó el 65 % y 56 % del total de especies obtenidas en granos de cebada en 2001 y 2002, respectivamente. La frecuencia de aislamiento de especies diferentes a F. graminearum varió dependiendo del ambiente y cultivar. En general, F. avenaceum, F. culmorum y F. poae fueron las especies más comúnmente obtenidas a partir de granos de trigo, mientras que F. equiseti, F. acuminatum y F. trincictum fueron las que se aislaron en menor frecuencia. En los granos de cebada, las especies más frecuentemente recuperadas luego de F. graminearum, fueron F. poae y F. equiseti. Otras especies recuperadas de cebada incluyeron a F. avenaceum, F. sambucinum, F. trincictum, F. semitectum y F. chlamydosporum. Todas las especies fueron patogénicas sobre espigas de trigo y cebada en los estudios de inoculación en el invernáculo, excepto F. semitectum en trigo. Las mayores severidades e incidencias de FE en las espigas de trigo y cebada se obtuvieron con los aislamientos de F. graminearum, seguidas por los de F. avenaceum y F. poae. Los cultivares previamente caracterizados como moderadamente resistentes a moderadamente susceptibles mostraron los menores niveles de incidencia y severidad de FE, porcentaje de granos infectados con Fusarium y granos infectados con F. graminearum. La información obtenida en este trabajo plantea preocupación en relación a la posible presencia de otras micotoxinas diferentes a DON (deoxinivalenol) en los granos de trigo y cebada en el país.
Palabras clave: Fusariosis de la espiga, Fusarium graminearum, Triticum aestivum, Hordeum vulgare, deoxinivalenol
Introduction
Fusarium head blight (FHB) has reemerged as a devastating disease of wheat and barley worldwide. Severe outbreaks have occurred in North America, Asia, Europe (Dubin et al., 1997 Windels, 2000) and in the southern cone of South America (de Galich, 1997; Díaz de Ackermann and Kohli, 1997). FHB is currently one of the main constraints to wheat and barley production in Uruguay.
Yield losses in wheat to FHB during the epidemic years of 1990, 1991, and 1993 have been estimated between 0.5 and 31 % (Díaz de Ackermann and Kohli, 1997). Yield losses for barley have not been thoroughly quantified, however FHB is considered important in both crops because of the contamination of grain with mycotoxins. Deoxynivalenol (DON or vomitoxin) is the most prevalent mycotoxin detected in Fusarium-infected grain in Uruguay (Piñeiro, 1997) and it is responsible for reducing the trading value of grain.
Fusarium head blight of wheat and barley in Uruguay is incited mainly by Fusarium graminearum (Schw.) [perfect stage Gibberella zeae (Schwabe) Petch] (Boasso, 1961; Pritsch, 1995; Pereyra and Stewart, 2001). Other Fusarium species also inciting FHB included, F. culmorum and F. poae in wheat grains (Stagno, 1980) and F. poae in barley (Pereyra and Stewart, 2001). However, there has not been a systematic survey of Fusarium species present in wheat and barley grain in different cultivars, locations and years.
The genus Fusarium includes a diversity of species that differ in their distribution according to climate, geography and host (Backhouse et al., 2001; BottalicoPerrone, 2002 and ; Doohan et al., 2003). Many of these Fusarium species produce a number of mycotoxins which pose a serious risk to human and animal health and generate great concern over the contamination of food products. The identification of the Fusarium species most prevalent in wheat and barley grains is critical to determine the risk of contamination by various toxins, and a necessary first step for effective management strategies. Therefore, the objectives of this study were i) to quantify the most prevalent Fusarium species in wheat and barley grain, ii) to quantify DON levels in wheat and barley grain, and iii) to assess the potential of the most prevalent Fusarium species to cause disease in wheat and barley plants.
Materials and methods
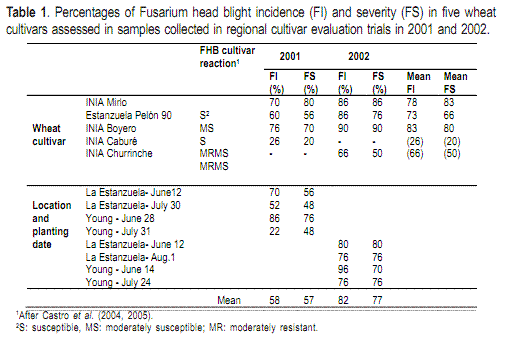
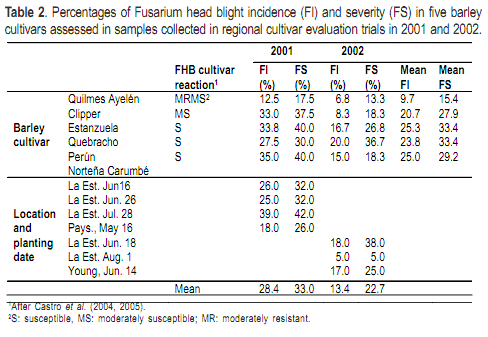
Grain samples (0.2 kg) of wheat and barley were collected from regional cultivar evaluation trials in 2001 and 2002. Five wheat and five barley cultivars that together comprised the bulk of the commercial hectarage in Uruguay were tested each year at La Estanzuela (Lat. 34º 20´S, long. 57º 41´W), Young (Lat. 32º 41´S, long. 57º 37´W) and Paysandú (Lat. 32º 19´S, long. 58º 05´W) and planted at different dates (Table 1 and 2).
Assessments of FHB were conducted on plots (5-m long, six rows, spacing 0.17 m) from which grains were obtained at the growth stages mid-milk to soft-dough (Zadoks growth stages 75-85, Zadoks et al., 1974). Wheat and barley plots were visually assessed based on a 0 to 100 % severity scale (percentage of symptomatic spikelets per spike) and a 0 to 100 % incidence scale (percentage of symptomatic spikes for the plot).
One hundred arbitrarily selected kernels per cultivar, planting date and location, were examined each year. The grain samples were surface disinfected by immersion in 0.5% sodium hypochlorite for 30 seconds, followed by immersion in 70 % ethanol for 15 seconds. The grain was then rinsed three times in sterile deionized water and blotted dry on sterile filter paper. Grain samples were plated onto pentachloronitrobenzene (PCNB) agar medium in Petri plates (Nash and Snyder, 1962). Twenty grains were placed per Petri plate, with five replicates, each plate represented a replicate. Plates were incubated at 20-22 °C with 12-h light and dark cycles provided by a 1:1 combination of cool white (F40/CW 25; Phillips, Sommerset, NJ) and blacklight (F40T12/BLB TL40W/08; Phillips, Sommerset, NJ) fluorescent lights for seven days. Subsequently, colonies growing with salmon to pink-white color were recorded as Fusarium species. Single conidial cultures were obtained by streaking a spore suspension made from the PCNB cultures onto water agar medium and transferring a single conidium, with a piece of surrounding agar. The proportion of Gibberella zeae colonies was determined by transferring 10 arbitrarily selected Fusarium spp. single conidial colonies to carnation-leaf piece agar (CLA) medium (Fischer et al., 1982) and potato-dextrose agar (PDA). Cultures on CLA were incubated at 20-22 °C with 12-h light and dark cycles for 15-20 days. The formation of bluish to black perithecia in CLA cultures indicated the presence of G. zeae. Fusarium colonies not forming perithecia were identified to species, based on procedures and descriptions outlined by Nelson et al. (1983) and Burgess et al. (1994).
Deoxynivalenol analyses were performed on all grain samples using a fluorometric quantitation method with Romer FluoroQuant®-DON (Romer Labs Inc., Union, MO) according to Malone et al. (1998).
Pathogenicity tests were conducted with single conidial isolates from different species and environments (locations/planting dates) collected in 2002. Tests were performed in the greenhouse with susceptible wheat line LE 2294 and susceptible barley cultivar Estanzuela Quebracho. Seeds were sown in plastic pots (150 by 150 mm, diam. by height) containing a 1:1:1 mixture of sand:soil:commercial substrate (Plantmax®, Eucatex, Sâo Paulo, Brazil). Five seeds were planted per pot and thinned to two plants per pot. Plants were maintained at 20 to 25 ºC with alternating 12-h light (high pressure sodium SON lamps, 400W, Phillips, Belgium) and dark periods.
Conidia were produced by incubating isolates on soybean medium (40 g of soybeans boiled in deionized water for 25 min, filtered through two layers of cheesecloth, adjusted to 1L; 15 g of agar) in Petri plates for 14 days. Conidia were harvested by adding 10 ml of sterile distilled water to each plate and gently scraping the culture with a sterile cover glass. Inoculum concentrations were adjusted to 2 x 104 macroconidia and microconidia per ml. One to two spikes per pot (five pots per isolate per plant species) were inoculated at mid-anthesis (Zadoks growth stages 65; Zadoks et al., 1974) using an airbrush (model VL3, Paasche Air Brush Company, Harwood Heights, IL) delivering 0.2 ml of inoculum per spike. Controls were mock-inoculated with sterile deionized water. Inoculated plants were incubated in a dew chamber at 20-22 ºC with a 12-h photoperiod and 100% relative humidity for 72 h. After incubation, plants were returned to the greenhouse and grown under the same conditions used prior to inoculation. Disease severity was evaluated 21 days after inoculation and expressed as the percentage of symptomatic spikelets per spike.
Fusarium species data were subjected to logistic regression using the SAS procedure GENMOD (version 8.1, SAS Institute Inc., Cary, NC). The results are presented as the likelihood ratio statistics of the Chi-square distribution. Pathogenicity data were subjected to analysis of variance using the GLM procedure of SAS (version 8.1, SAS Institute Inc., Cary, NC). Where the F ratio was significant (P<0.05), differences among treatment means were separated using Fischer’s protected least significant differences (LSD) test. Data for FHB severity, FHB incidence, Fusarium spp. and F. graminearum isolation frequency, and DON concentration were subjected to Pearson correlation analyses to measure associations. Correlation was not performed with other Fusarium species because they were recovered at very low frequencies.
Results
Disease assessments
The percentage FHB incidence (FI) and severity (FS) in the wheat cultivars were generally higher than the FI and FS in barley (Tables 1 and 2). The mean FI and FS in wheat, across cultivars and environments, were generally higher in 2002 than in 2001 (Table 1). The lowest FI and FS in wheat were obtained in cultivars INIA Caburé in 2001 and INIA Churrinche in 2002. In 2001, the highest FI and FS were observed in Young in plots planted on June 28. In 2002, all environments examined had high FI and FS and differences among sites or planting dates were not apparent.
The mean FI and FS for barley, across cultivars and environments, were higher in 2001 compared to 2002 (Table 2). In both years, the lowest disease incidences and severities were observed in Quilmes Ayelén. In 2001, cultivars other than Quilmes Ayelén had similar high incidences and severities. For this year, the average FI and FS at Paysandú was lower than the average FI and FS for any of the planting dates at La Estanzuela. In 2002 the mean FI and FS values for barley were lowest at the late planting date (August 1) in La Estanzuela.
Isolation frequency of Fusarium species and DON content
Wheat grain colonization by Fusarium species averaged 32 % in 2001 and 51 % in 2002. Fusarium was isolated from 31 % and 18 % of barley grains in 2001 and 2002, respectively.
There was a significant effect of wheat cultivar in 2001 on the frequency of isolation of Fusarium species (Table 3). INIA Caburé had the lowest frequency of isolation of Fusarium species in 2001. Although the differences were not significant, in 2001, early planting dates had higher Fusarium isolation frequencies than late planting dates, irrespective of location. In 2002, there was a significant effect of environment on the frequency of Fusarium species recovered. The late planting date at La Estanzuela and the early planting date at Young had the lowest percentages of Fusarium species recovered from wheat. The cultivar by environment interaction was significant in 2001 and 2002.
In both years, the recovery of Fusarium species from barley samples was significantly influenced by cultivar (Table 4). The cultivar Quilmes Ayelén consistently had the lowest frequencies of Fusarium-infested grain. Perún had the highest frequency of Fusarium-infested grain in 2001, while Estanzuela Quebracho had the highest infestation in 2002. In 2001, grain at Paysandú had the lowest Fusarium isolation frequency. In 2002, a significantly lower percentage of Fusarium-infested grain was obtained in Young. The interaction between cultivars and environment was significant for Fusarium-infested barley grain in both 2001 and 2002.
Seven Fusarium species were isolated from the wheat grains and nine from the sampled barley grains (Fig. 1). The Fusarium species F. graminearum, F. avenaceum, F. poae, F. equiseti, F. acuminatum, and F. trincictum were found in grains of both crops. Fusarium culmorum was only recovered from wheat, while F. sambucinum, F. semitectum, and F. chlamydosporum were only isolated from barley. In 2002, F. acuminatum and F. trincictum were recovered from wheat grains although they had not been recovered from wheat sampled the previous year. Similarly, F. semitectum and F. chamydosporum were isolated for the first time from barley grains in 2002.
Fusarium graminearum was the most frequently recovered species in both years from wheat and barley samples in all environments sampled (Tables 3 and 4; Fig.1). Fusarium graminearum comprised 65 % and 56 % of the Fusarium species recovered from barley grains in 2001 and 2002 (Fig. 1). The isolation frequencies of F. graminearum in both crops and years followed the same pattern as the isolation frequencies of all Fusarium species, regardless of cultivars and environment. The frequencies, with which Fusarium species other than F. graminearum were recovered, varied depending on both the environment and host cultivar (Tables 3 and 4).
Fusarium avenaceum was the second most commonly recovered species from wheat grains (Fig. 1). The frequency with which F. avenaceum was isolated varied depending on the wheat cultivar and environment. Fusarium avenaceum was more frequently recovered from INIA Boyero in both 2001 and 2002 and from INIA Churrinche in 2002. This species was isolated most frequently from wheat grains from the 2001 late planting date at La Estanzuela and from the early planting date at La Estanzuela and the late planting date at Young in 2002 (Table 3).
Fusarium culmorum was never isolated from more than 6 % of the wheat grain samples examined and was not isolated from Estanzuela Pelón in 2001, or from INIA Churrinche in 2002. Fusarium culmorum was not isolated from late planted grain grown at La Estanzuela in either 2001 or 2002. Fusarium culmorum was not recovered from INIA Boyero or from any cultivar at the late planting date at La Estanzuela in 2001. The remaining Fusarium species recovered from wheat grains represented less than 4 % on average, except F. trincictum which was isolated from 6% of wheat grain in 2002 (Fig. 1), principally from the wheat cultivar INIA Churrinche (Table 3).
Fusarium poae was the second most frequently recovered Fusarium species from barley (Fig. 1). In Paysandú, F. poae was recovered at a higher frequency than F. graminearum. Fusarium poae was recovered from all barley cultivars and environments, but was most frequently isolated in 2001 from Clipper and in 2002 from Estanzuela Quebracho. In both years, F. poae was most frequently recovered from barley grains sampled from the late planting dates at La Estanzuela (Table 4).
Fusarium equiseti represented the third most frequently isolated species from barley grains in both years (Fig. 1). It was isolated from all environments and all cultivars, except from Quilmes Ayelén (Table 4). The other Fusarium species recovered were found in less than 5 % of the barley grains tested and recovery varied according to the cultivar and environment (Fig. 1, Table 4).
Deoxynivalenol was detected in all wheat and barley samples tested in 2001 and 2002 (Tables 3 and 4). The DON content of individual samples ranged from 1.3 to 13.0 ppm in 2001 and 1.6 to 19.0 ppm in 2002 for wheat and from 1.8 to 34.0 ppm in 2001 and 0.5 to 9.1 ppm in 2002 for barley. The highest levels of DON were consistently obtained from the wheat cultivar INIA Boyero and the barley cultivars Perún and Estanzuela Quebracho. The highest mean DON content in wheat was observed in the samples collected at the early planting date at La Estanzuela in 2001 and at Young in 2002 (Table 3). Barley grains collected in La Estanzuela in 2001 and in Young in 2002 had the highest mean DON concentrations for barley (Table 4).
Pathogenicity tests
All Fusarium isolates tested caused visible symptoms of FHB in wheat and barley spikes, except for the isolates of F. sambucinum and F. semitectum on wheat (Table 5). The F. graminearum isolates generated greater FHB severities in wheat and barley plants than the other Fusarium isolates tested.
Fusarium isolates varied significantly in disease severity on wheat. Fusarium graminearum isolated from wheat caused significantly higher FHB severity on wheat spikes than other F. graminearum isolates tested. There was a trend for obtaining higher mean FHB severities for the F. graminearum isolates isolated from barley inoculated on barley spikes when compared with the rest of the species tested, however differences were not significant. Spikes mock-inoculated with water did not develop any symptoms of FHB.
Correlation tests
The incidence and severity of FHB assessed at the experimental sites were positively correlated with the percentage of grains infested with Fusarium species, the percentage of grains infested with F. graminearum, and DON content (Table 6). The highest correlation coefficients were between Fusarium species and F. graminearum, FHB incidence and isolation frequency of Fusarium species and between FHB severity and isolation frequency of Fusarium species in 2001 for both wheat and barley (Table 6). There was also a positive correlation between the F. graminearum isolation frequency and the DON content in both crops, although this was only significant for barley sampled in 2001. The correlation coefficients among the other variables examined were very low and were not significant (Table 6).
Discussion
FHB occurred widely in the wheat and barley production areas of Uruguay in 2001 and 2002, and disease incidence and severity varied according to wheat and barley genotype and environment. The information obtained in this study examining the most popular wheat and barley cultivars in that period, grown at different locations and planting dates, provided a means to characterize the 2001 and 2002 epidemics in terms of the prevalent Fusarium species, disease incidence and severity, and DON accumulation.
In general, cultivars characterized as moderately resistant to moderately susceptible, like INIA Caburé, INIA Churrinche (in wheat) and Quilmes Ayelén in barley (Castro et al., 2004; Castro et al., 2005), had the lowest incidences and severities of FHB, percentages of Fusarium-infested grain and F. graminearum-infested grain. Conversely, susceptible cultivars, such as INIA Boyero (wheat) and Perún (barley), had the highest level of disease and DON content of harvested grain. Given that the barley cultivars Quilmes Ayelén and Perún had similar heading dates in all environments tested, Quilmes Ayelén may have some resistance to FHB which resulted in lower FHB disease ratings and levels of F. graminearum-infested grains. The wheat cultivar INIA Mirlo and barley cultivar Norteña Carumbé had consistently high levels of disease and F. graminearum-infested grain, however, they also had the lowest levels of DON in both years. Mechanisms of resistance which prevent toxin accumulation in wheat have been proposed by Mesterházy (1995). Further research is needed to corroborate if any of the cultivars examined in this study have resistance with a specific effect on DON accumulation.
Climatic conditions in relation to heading/flowering dates and grain filling period (Pereyra, 2005) might explain some of the variation in FHB incidence, severity, and the proportions of Fusarium species recovered among the different environments tested. Climatic influences on FHB have been reported by Langseth et al. (1995), Salas et al. (1999), and Doohan et al. (2003). Ascospore production and spike infection are favored by moist conditions, especially in wheat during flowering and in barley between heading and harvest (Bushnell et al., 2003; Doohan et al., 2003). In 2001, early plantings (June) at La Estanzuela and Young had the highest levels of FHB, F. graminearum-infested grains and DON. This was probably a consequence of the wheat and barley spikes being exposed to longer periods favorable for disease development as plots received rainfall at and after flowering. Favorable conditions for infection with frequent rains greater than 0.2 mm occurred in late September at La Estanzuela and Young. Barley planted at Paysandú in 2001 experienced fewer rain events as it was harvested earlier (mid-November) than the rest of the 2001 trials. This may explain the low level of FHB in barley at this site. In 2002, favorable conditions for FHB occurred in all tested environments. Barley planted at La Estanzuela in August 2002 had the lowest levels of FHB at milk grain stage, however barley at La Estanzuela also had the highest percentage of Fusarium-infested grains in 2002.
The results from isolation and identification of Fusarium species in wheat and barley grains clearly confirmed that the main species associated with FHB in Uruguay is F. graminearum as suggested by previous studies (Boasso, 1961; Pritsch, 1995; Pereyra and Stewart, 2001). Similar results are reported for North America (McMullen et al., 1997; Salas et al., 1999; Clear and Patrick, 2000), some regions of Europe (Parry et al., 1995) and other countries in South America (Reis, 1988; Lori et al., 2003). Fusarium graminearum is widespread in the southern cone of South America and has been isolated from a wide range of hosts (Reis, 1988; Pereyra and Dill-Macky, 2008).
Subsequent studies focusing on the phylogenetic lineages [or phylogenetic species according to O´Donnell et al. (2004)] of F. graminearum sensu lato have shown that phylogenetic lineage 7 (F. graminearum sensu O´Donnell) was the most common lineage isolated from wheat grains obtained from farmers fields in Uruguay during 2001, 2002, and 2003 (Pereyra et al., 2006). Other lineages found included lineage 1 (F. austroamericanum) and lineage 8 (F. cortaderiae). Presence of these lineages have already been cited in the Southern Cone of South America by several authors (Leslie and Bowden, 2005; O´Donnell et al., 2004; Zeller et al., 2002; Zeller et al., 2003).
In terms of toxin production, strains of F. graminearum sensu lato have been classified as either DON-chemotype IA, producing DON and 3-acetyl-DON and found especially in warmer cereal growing regions, or DON-chemotype IB, producing DON and 15-acetyl-DON in slightly cooler regions (Miller et al., 1991). Uruguayan isolates of F. graminearum belong to chemotype IB (Piñeiro, 1997; Pereyra et al., 2006). Isolates of Fusarium graminearum from Uruguay also characteristically produce zearalenone (ZEA) (Piñeiro, 1997).
Fusarium avenaceum was the second most common species found in wheat grains while either F. culmorum or F. poae was the third most common species, depending on the year. These three species are most often found in cooler production areas (Backhouse et al., 2001; Bottalico and Perrone, 2002) and this might explain the higher incidence of these species at the early planting dates in the southern region of Uruguay (La Estanzuela) where cooler conditions occurred at heading/flowering. Fusarium culmorum is known to produce DON (Thrane, 2001) and thus would likely contribute to DON levels in wheat grains when present. Fusarium avenaceum produces mainly moniliformin (MON), while F. poae produces diacetoxyscirpenol (DAS), fusarenon (FUS), nivalenol (NIV), T-2 and HT-2 (Thrane, 2001; Bottalico and Perrone, 2002). Among the other less frequently recovered species in wheat were F. equiseti (producing DAS and ZEA), F. acuminatum (producing T2), and F. trincictum (producing MON) (Thrane, 2001; Bottalico and Perrone, 2002).
Fusarium poae was the second most prevalent species in barley grains. Under Uruguayan conditions, infection by F. poae usually occurs at boot stage (G. S. 45-49; Zadoks et al., 1974), infecting the spike through the flag leaf sheath in late-August or early-September when lower temperatures are generally more favorable for disease development. The pathogenicity and increasingly wide distribution of F. poae in barley should not be overlooked by barley breeding programs when screening lines for resistance to FHB.
A greater range of Fusarium species was found in barley compared to wheat, including the species F. sambucinum, F. semitectum, and F. chlamydosporum, which were not isolated from any wheat samples. All species recovered from grain caused FHB symptoms, except F. sambucinum and F. semitectum inoculated onto wheat. The highest levels of FHB occurred in spikes of wheat and barley inoculated with F. graminearum. The prevalence of individual species and their relative frequencies within the FHB pathogen complex may fluctuate in response to host factors (host species, cultivar) and climate, as associated with location, (temperature, rainfall patterns, humidity, soil type) (Backhouse et al., 2001; Doohan et al., 2003; Waalwijk et al., 2003; Shah et al., 2005). This study demonstrated that both pathogenic (i.e. F. graminearum, F. avenaceum and F. poae) and opportunistic Fusarium species (i.e. F. equiseti) may co-exist in a contaminated crop. These species mixtures may generate unique mycotoxigenic profiles in the contaminated grain.
The highest correlation coefficients in this study between the percentage of Fusarium-infested grains and the percentage of F. graminearum-infested grains were not surprising as F. graminearum was the predominant species in the Fusarium complex. The low correlation coefficients between field FHB incidence and DON, as well as between field FHB severity and DON, in both crops might be explained by many factors as reported by Blaney and Dodman (2002), Cromey et al. (2002) and Mirocha et al. (1989). Results from this study suggest that the presence of non-DON producing species of Fusarium provides an additional source of variation to the estimation of DON content in grains based on FHB symptoms. Furthermore, the presence of Cochliobolus sativus, a common pathogen of barley in Uruguay (Pereyra, 1996) causing lesions similar to FHB in the spikes, was significant in both years.
The results from this study have also shown that several potentially important toxin-producing Fusarium species are common under natural conditions in wheat and barley grains in Uruguay.
References
Backhouse, D., Burgess, L. W., and Summerell, B. A. 2001. Biogeography of Fusarium. Pages 122-136 in: 2001 Fusarium Paul E. Nelson Memorial Symposium. B. A. Summerell, J. F. Leslie, D. Backhouse, P. Bryden, and L. W. Burgess, eds. The American Phytopathological Society Press. St. Paul, MN.
Blaney, B.J. and Dodman, R.L. 2002. Production of zearalenone, deoxynivalenol, nivalenol, and acetylated derivatives by Australian isolates of Fusarium graminearum and F. pseudograminearum in relation to source and culturing conditions, Aust. J. Agric. Res. 53:1317–1326.
Boasso, C. 1961. Estado fitosanitario de los cultivos de trigo de la reciente cosecha. Boletín Informativo 854, pag.7.
Bottalico, A. and Perrone, G. 2002. Toxigenic Fusarium species and mycotoxins associated with head blight in small-grain cereals in Europe. Eur. J. Plant Pathol. 108:611-624.
Burgess, L. W., Summerell, B. A., Bullock, S., Gott, K. P., and Backhouse, D. 1994. Laboratory Manual for Fusarium Research. Third edition. University of Sydney, Sydney, Australia. 133p.
Bushnell, W. R., Hazen, B. E., and Pritsch, C. 2003. Histology and physiology of Fusarium head blight of wheat and barley. Pages 44-83 in: Fusarium head blight of wheat and barley. K. Leonard and W. Bushnell, eds. The American Phytopathological Society Press. St. Paul, MN.
Castro, M., Pereyra, S., Germán, S., Vázquez, D. 2004. Resultados experimentales de evaluación de trigos y cebadas cerveceras de los 3 últimos años para el registro nacional de cultivares. Período 2003. INASE-INIA. 30p.
Castro, M., Pereyra, S., Germán, S., Vázquez, D. 2005. Resultados experimentales de evaluación de trigos y cebadas cerveceras de los 3 últimos años para el registro nacional de cultivares. Período 2002. INASE-INIA. 27p.
Clear, R. M. and Patrick, S. K. 2000. Fusarium head blight pathogens from fusarium-damaged kernels of wheat in western Canada, 1993 to 1998. Can. J. Plant Pathol. 22:51-60.
Cromey, M.G., Shorter, S.C., Lauren, D.R., and Sinclair, K.I. 2002. Cultivar and crop management influences on Fusarium head blight and mycotoxins in spring wheat (Triticum aestivum) in New Zealand. N. Z. J. Crop Horticultural Sci. 30:235–247.
de Galich, M. T. 1997. Fusarium head blight of wheat in Argentina. Pages 19-28 in: Fusarium head scab: Global status and future prospects. H. J. Dubin, L. Gilchrist, J. Reeves, and A. McNab, eds. CIMMYT, DF, Mexico.
Díaz de Ackermann, M. and Kohli, M. 1997. Research on Fusarium head blight in Uruguay. Pages 13-18 in: Fusarium head scab: Global status and future prospects. H. J. Dubin, L. Gilchrist, J. Reeves, and A. McNab, eds. CIMMYT, DF, Mexico.
Doohan, F. M., Brennan, J. and Cooke, B. M. 2003. Influence of climatic factors on Fusarium species pathogenic to cereals. European J. Plant Pathol. 109:755-768.
Dubin, H. J., Gilchrist, L., Reeves, J., and McNab, A. (eds.) 1997. Fusarium head scab: Global status and future prospects. CIMMYT, DF, Mexico. 130 p.
Fischer, N. L., Burgess, L. W., Tousson, T. A., and Nelson, P. E. 1982. Carnation leaves as a substrate for preserving cultures of Fusarium species. Phytopathology 72:151-153.
Langseth, W., Hoie, R., and Gullord, M. 1995. The influence of cultivars, location and climate on deoxynivalenol contamination in Norwegian oats 1985-1990. Acta Agric. Scand. 45:63-67.
Leslie, J. F. and Bowden, R. L. 2005. Field populations of Gibberella zeae. p.166. in: Canty, S.M.; Boring, T.; Wardwell, J., Siler, L. and Ward, R. W. (eds.) Proceedings of the National Fusarium Head blight Forum; 2005 Dec. 11-13; Milwakee, WI. Michigan State University, East Lansing, MI.
Lori, G. A., Sistema, M. N., Haidukowski, M. and Rizzo, I. 2003. Fusarium graminearum and deoxynivalenol contamination in the durum wheat area in Argentina. Microbiol. Res. 158:29-35.
Malone, B. R.; Humphrey, C. W.; Romer, T.R.; Richard, J. L. 1998. One-step solid-phase extraction cleanup and fluorometric analysis of deoxynivalenol in grains. Journal of AOAC International. 81: 448-452.
McMullen, M., Jones, R., and Gallenberg, D. 1997. Scab of wheat and barley: A re-emerging disease of devastating impact. Plant Dis. 81:1340-1348.
Mesterházy, Á. 1995. Types and components of resistance to Fusarium head blight of wheat. Plant Breed. 114:377-386.
Miller, J. D., Greenhalgh, R., Wang, Y. Z., and Lu, M. 1991. Trichothecene chemotype of three Fusarium species. Mycologia 83:121-130.
Mirocha, C.J., Abbas, H.K., Windels, C.E., and Xie, W. 1989. Variation in deoxynivalenol, 15-acetyldeoxynivalenol, 3-acetyldeoxynivalenol and zearalenone production by Fusarium graminearum isolates, Appl. Environ. Microbiol. 55:1315–1316.
Nash, S. M. and Snyder, W. C. 1962. Quantitative estimations by plate counts of propagules of the bean root rot Fusarium in field soils. Phytopathology 52:567-572.
Nelson, P. A., Tousson, T. A., and Marasas, W. F. O. 1983. Fusarium species: An Illustrated Manual for Identification. The Pannsylvania State University Press. University Park. 193 p.
O´Donnell, K.; Ward, T. J.; Geiser, D. M.; Kistler, H. C. and Aoki, T. 2004. Genealogical concordance between the mating type locus and seven other nuclear genes supports formal recognition of nine phylogenetically distinct species within the Fusarium graminearum clade. Fungal Genet. Biol. 41:600-623.
Parry, D. W., Jenkinson, P., and McLeod, L. 1995. Fusarium ear blight (scab) in small grain cereals – a review. Plant Pathology 44:207-238.
Pereyra, S. 1996. Enfermedades de cebada: identificación, epidemiología y estrategias de manejo. Pages 105-123 in: Curso de actualización para el manejo de enfermedades en cereales de invierno y pasturas. Serie técnica 74. M. Díaz ed. INIA. Colonia. Uruguay.
Pereyra, S. A. 2005. Epidemiological and ecological studies on pathogenic Fusaria causing Fusarium head blight of wheat and barley in Uruguay. PhD Dissertation. University of Minnesota. St. Paul.131p.
Pereyra, S. and Stewart, S. 2001. Investigación en fusariosis de la espiga de cebada en Uruguay. Pages 41-68. in: XXI Reuniao Anual de Pesquisa de cevada Anais e ata. E. Minella, ed. EMBRAPA, Passo Fundo, Brazil.
Pereyra, S.A., Vero, S., Garmendia, G., Cabrera, M., Pianzolla, M. J. 2006. Diversity of Fungal Populations Associated with Fusarium Head Blight in Uruguay. in: Ban, T., J.M. Lewis, and E.E. Phipps (eds.) The Global Fusarium Initiative for International Collaboration: A Strategic Planning Workshop held at CIMMYT, El Batán, Mexico; March 14 - 17, 2006. México, D.F.: CIMMYT., pp. 35-41.
Pereyra, S.A. and Dill-Macky, R. 2008. Colonization of the Residues of Diverse Plant Species by Gibberella zeae and their Contribution to Fusarium Head Blight Inoculum.. Plant Dis. 92:800-807.
Piñeiro, M. 1997. Fusarium toxins in Uruguayan wheat. Pages 125-128 in: Fusarium head scab: Global status and future prospects. H. J. Dubin, L. Gilchrist, J. Reeves, and A. McNab, eds. CIMMYT, DF, Mexico.
Pritsch, C. 1995. Variabilidad patogénica en Fusarium spp. agente causal del golpe blanco del trigo. FPTA-INIA. Informe final 79p.
Reis, E. M. 1988. Doenças do trigo III. Giberela. Segunda ediçao. Sao Paulo, Brazil. 12p.
Salas, B., Steffenson, B. J., Casper, H. H., Tacke, B., Prom, L. K., Fetch, T. G. and Schwarz, P. B. 1999. Fusarium species pathogenic to barley and their associated mycotoxins. Plant. Dis. 83:667-674.
Shah, D. A., Pucci, N., and Infantino, A. 2005. Regional and varietal differences in the risk of wheat seed infection by fungal species associated with Fusarium head blight in Italy. Eur. J. Plant Pathol. 112:13-21.
Stagno, J. P. 1980. Enfemedades transmitidas por la semilla de trigo. Boletin Semagro 3:30-33.
Thrane, U. 2001. Developments in the taxonomy of Fusarium species based on secondary metabolites. Pages 29-49 in: Fusarium. Paul E. Nelson memorial symposium. B. E. Summerell, J. F. Leslie, D. Backhouse, W. L. Bryden, L. W. Burgess, eds. The American Phytopathological Society Press. St. Paul, MN.
Waalwijk, C., Kastelein, P., de Vries, I., Kerenyi, Z., van der Lee, T., Hesselink, T., Kohl, J., and Kema, G. 2003. Major changes in Fusarium spp. in wheat in the Netherlands. Eur. J. Plant Pathol. 109:743-754.
Windels, C. E. 2000. Economic and social impacts of Fusarium head blight: Changing farms and rural communities in the Northern great plains. Phytopathology 90:17-21.
Zadoks, J.C., Chang, T.T., and Konzac, C.F. 1974. A decimal code for the growth stages of cereals. Weed Res. 14:415-421.
Zeller, K.A.; Vargas, J.I.; Valdovinos-Ponce, G.; Leslie, J.F. and Bowden. R.L. 2002. Population genetic differentiation and lineage composition among Gibberella zeae (Fusarium graminearum) in North and South America. p. 188 in: Canty, S. M.; Lewis, J.; Siler, L. and Ward, R. W. (eds.) Proceedings of the National Fusarium Head blight Forum; 2002 Dec. 7-9; Erlanger, KY. Michigan State University, East Lansing, MI.
Zeller, K.A.; Vargas, J.I.; Valdovinos-Ponce, G.; Leslie, J.F. and Bowden. R.L. 2003. Population genetic differentiation and lineage composition among Gibberella zeae in North and South America. Fungal Genet. Newsl. 50:446.