Servicios Personalizados
Revista
Articulo
Links relacionados
Compartir
Odontoestomatología
versión impresa ISSN 0797-0374versión On-line ISSN 1688-9339
Odontoestomatología vol.21 no.34 Montevideo dic. 2019 Epub 01-Dic-2019
https://doi.org/10.22592/ode2019n34a3
Research
Immunoexpression of biomarkers Bax, Bcl-2, CD-138, H3, Ki-67, MCM3 and p53 in oral lichen planus
1Área de Patología Molecular Estomatológica. Facultad de Odontología, Universidad de la República, Uruguay.
2Cátedra de Anatomía Patológica A. Facultad de Odontología, Universidad Nacional de Córdoba, Argentina.
3Cátedra de Histología. Facultad de Odontología, Universidad de la República, Uruguay.
4Departamento de Sistemas Biológicos. División de Ciencias de la Salud. Universidad Autónoma Metropolitana, México.
5Área de Patología Molecular Estomatológica. Facultad de Odontología, Universidad de la República, Uruguay.
This study aims to establish an association of the expression of specific biomarkers in oral lichen planus to understand its biological behavior. Materials and methods: An immunohistochemistry study was conducted in 40 cases of oral lichen planus against BAX, BCL-2, CD-138, Histone 3, Ki-67, MCM3 and p53 at the Molecular Pathology Area of the School of Dentistry, UDELAR, Uruguay. Results: A greater expression of BAX was detected compared to BCL-2, suggesting a pro‑apoptotic behavior, supported by the absence of p53 expression. MCM3 expression was more sensitive than Ki-67, considering proliferation alterations. CD-138 had a more intense and uniform expression, determining fewer intercellular adhesion alterations. Conclusions The expression of the proteins studied suggests an alteration in proliferative and apoptotic mechanisms, associated with a pathological behavior of the oral mucosa.
Key words: oral lichen planus; apoptosis; proliferation
El objetivo del presente trabajo fue determinar la expresión de diversos biomarcadores moleculares en liquen plano oral para ayudar a comprender su conducta biológica. Materiales y métodos: Se realizó un estudio inmunohistoquímico en 40 casos de liquen plano oral contra BAX, BCL-2, CD-138, Histona 3, Ki-67, MCM3 y p53, en el Área de Patología Molecular Estomatológica de la Facultad de Odontología, UDELAR, Uruguay. Resultados: Se observó mayor expresión de BAX en contraposición con BCL-2, sugiriendo un comportamiento proapoptótico, respaldado a su vez por la ausencia de expresión de p53. La expresión de los marcadores de proliferación celular fue en todo el tejido lesional observado, sugiriendo así alteraciones de la proliferación. CD-138 se expresó de manera intensa y uniforme, determinando una baja alteración de las uniones intercelulares para estos casos. Conclusiones: La alteración en la expresión de las proteínas estudiadas sugiere un trastorno en los mecanismos proliferativos y apoptóticos, los cuales se asocian con una conducta patológica de la mucosa oral.
Palabras clave: liquen plano oral; apoptosis; proliferación
Introduction
Lichen planus is an immune-mediated inflammatory disease of the stratified squamous epithelium that affects the skin and mucosa1. It was first described by Erasmus Wilson in 1869. Etymologically, “lichen planus” is derived from the Greek word leichen, “tree moss”, and the Latin planus, “flat”, due to the appearance of the lesions2. It affects 0.2-1.9% of the population and is mostly found in people aged between 30 and 703. While cutaneous lichen is self-limited, oral lichen planus (OLP) is chronic, rarely undergoes spontaneous remission and is considered potentially malignant by the World Health Organization4, with the risk of malignant transformation into oral squamous cell carcinoma being less than 1%5.
As for its clinical features, the most common location for lesions is the buccal mucosa, followed by the tongue (lateral borders), the dorsum of the tongue and the gingiva. OLP can have different clinical presentations, among which are reticular lichen planus, characterized by a pattern of whitish striae (keratinization) 0.1 to 2 mm wide; atrophic lichen planus, which shows redness and thinning of the epithelium; ulcerated lichen planus defined by loss of epithelial surface, with formation of irregular ulcers that are covered with fibrin, surrounded by atrophic areas; plaque-like lichen planus with solid areas of keratinization (mainly in the tongue or buccal mucosa); desquamative gingivitis, which affects the attached gingiva, which appears inflamed, smooth and shiny; bullous lichen planus is found when there is a separation of the epithelium after the loss of basal cells5-7.
As for its histopathology, OLP can manifest with various alterations such as hyperkeratosis (ortho- or parakeratosis), acanthosis, granulosis, spongiosis, colloid bodies, lymphocytic exocytosis and epithelial atrophy, with the essential alterations being vacuolar degeneration of the basal layer of the epithelium and the underlying band-like inflammatory infiltrate8.
The etiopathogenesis of OLP has not been completely elucidated, but it is thought to be a cytotoxic T cell (CD8+)-mediated autoimmune reaction against basal keratinocytes, induced by an antigenic change in the skin or mucosa9. The combination of specific and nonspecific mechanisms seem to cause the accumulation of T lymphocytes in the lamina propria which underlies the epithelium, breaks in the basal lamina, the migration of intraepithelial T lymphocytes and keratinocyte apoptosis10. The altered antigens of basal keratinocytes are recognized and presented by Langerhans cells to CD4+ T lymphocytes locally or during passage through the lymph nodes through the HLA-II (class II Human Leukocyte Antigen), triggering the clonal expansion. After the T cells have become activated, they differentiate into two effector subtypes: type 1 T cells (Th1) which produce TNFγ (Tumor Necrosis Factor γ) and type 2 T cells (Th2) which produce interleukins (IL): IL-4, IL-5, IL-6, IL-10 and IL-1311. TNFγ stimulates the accelerated proliferation of basal keratinocytes and the subsequent accumulation of surface keratin that clinically manifests as hyperkeratosis; under this stimulus, cell layers in different strata increase, and epithelial desquamation becomes slower. The intense basal proliferation causes intercellular alterations, which are perceived by intraepithelial CD8+ T lymphocytes through HLA I expression by basal keratinocytes, and apoptosis is activated in the basal layer12.
The mutations and molecular alterations present in OLP might play a critical role in neoplastic transformation13.
According to Fitzpatrick, erosive OLP shows a higher risk of neoplastic transformation14.
OLP is commonly diagnosed using the WHO's clinical and pathological criteria, which includes the presence of bilateral, mostly symmetrical lesions; white reticular-papular striation; erosive, atrophic, bullous and plaque-type lesions in the presence of white striae elsewhere in the oral cavity. Histopathological criteria are based on the presence of a band-like juxtaepithelial infiltrate, consisting mainly of lymphocytes; signs of liquefaction degeneration in the basal cell layer, and absence of epithelial dysplasia15.
Treatment of OLP usually entails the application of topical and/or systemic corticosteroids5.
The study of several biomarkers, for cell proliferation (Ki-67, MCM3, Histone 3), for tumor suppression (p53) and for the regulation of the apoptosis process (BCL-2, BAX) can help establish a possible association between the expression of these proteins and the pathological behavior of OLP. Therefore, the aim of this study was to determine and find a morphological correlation between the presence of these proteins and OLP to generate information that helps us understand the biological behavior of these lesions.
Materials and methods
We included 40 cases of OLP from samples from the Pathology Department of Universidad Autónoma Metropolitana (UAM, Mexico City, Mexico) and Universidad de Córdoba, Argentina, diagnosed during the period ranging from 2008 to the beginning of 2013. The specimens were preserved in paraffin blocks. Seven sections of the tissue were cut for the immunohistochemistry technique and one section for hematoxylin and eosin staining. The morphological study was conducted by two pathologists experienced in OLP, taking into account predetermined histopathological parameters and the clinical features of the lesion16.
Immunohistochemical studies were conducted at the Laboratory of Molecular Pathology in Stomatology, of the School of Dentistry, Universidad de la República, (UDELAR, Montevideo, Uruguay). This study was approved by the Ethics Committee of the School of Dentistry, UDELAR, file number: 120/16.
We cut 4 µm sections and mounted them on poly-L-lysine coated slides. The sections were then deparaffinized in a stove at 60°C for 30 minutes and subsequently left in xylol for 5 minutes. The sections were hydrated in a series of decreasing concentrations of absolute alcohols (absolute, 90%, 80%, 70% and 50%) and rinsed with distilled water. The antigen retrieval technique was used to unmask epitopes with a pH 6 sodium citrate solution in a pressure cooker in microwave oven on full power at 750 W. Endogenous peroxidases were blocked with hydrogen peroxide at 0.9%, followed by washes with distilled water and a pH 7.4 phosphate-buffered saline (PBS) solution. Primary antibodies were incubated for 60 minutes against: p53 (dilution: 1:200, Bio SB/D07), MCM3 (dilution: 1:100, Leica/DCS-141.1), Ki-67 (dilution: 1: 100, DAKO/MIB1), CD-138 (dilution: 1: 100, DAKO/MI15), BCL-2 (dilution: 1:100, Biocare/100/D5), BAX (dilution: 1:200, DAKO Corp, Carpinteria, CA, USA, Policlonal), histone H3 (dilution: 1:50, phosphohistone H3 Genetex 300095). The sections were then incubated with the second biotinylated anti-mouse/anti-rabbit antibody and with the streptavidine/peroxidasa complex (LSA-B + Labeled streptavidin-biotin, Dako Corporation, Carpinteria CA, USA) for 30 minutes each. The products of the reaction were visualized with substrate 3,3’-diaminobenzidine-H2O2 (Dako Corporation, Carpinteria, CA, USA). Various tissue punches were used as positive controls; as negative controls the incubation with the primary antibodies was omitted.
Cytoplasmic and/or membrane quantification was done visually using an optical microscope (Eclipse CI-L, Nikon, Japan) with a 40x magnification, taking into account the following semi-quantitative scale: 0-4% "negative staining"; 5-25% are considered a "weak staining"; 26-50% were "moderate staining" and 51-100% corresponds to a "strong staining" of cells. Nuclei were quantified by dividing the total number of positive cells by the total number of cells present in the field being analyzed and multiplying by 100 to obtain a percentage. Microphotographs of all markers were taken using a digital camera (Olympus C-7070) in three selected fields in which larger areas of epithelial damage caused by the inflammatory response (corresponding to the basal layer and the subepithelial infiltrate) were found.
Results were compared subjectively against unaltered healthy tissue from the analyzed sample when the sample size allowed. This was used as an internal control. When no tissue was available for comparison, we used healthy tissue from another mucosa sample as reference.
Given that the aim of the study was to evaluate the presence of the relevant proteins at the morphological level, no clinical features were associated with this study.
Results
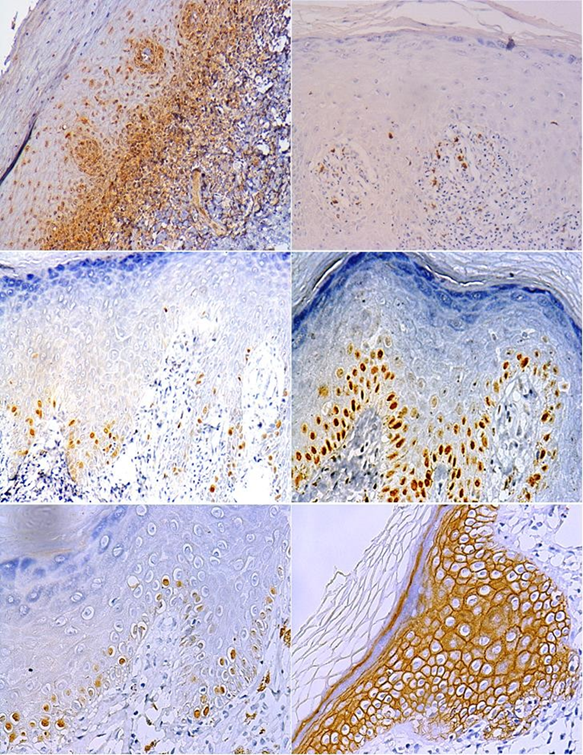
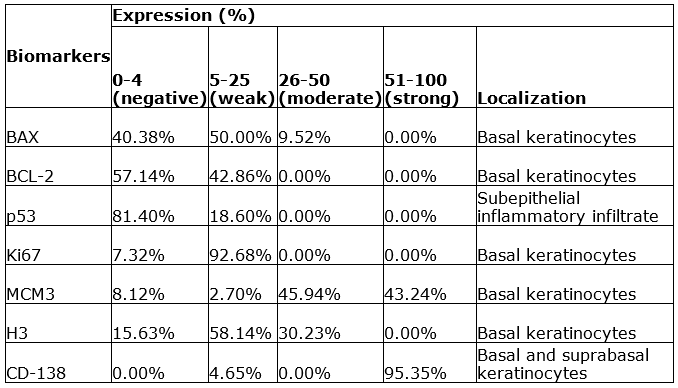
The expression of apoptosis-related markers, as is the case of BAX, was predominantly weak for 50% of samples, localized in the basal layer of the epithelium, and found in the cytoplasm of basal keratinocytes (Fig. 1. A), while BCL-2 showed weak cytoplasmic expression with 42.86% of cases, in suprabasal layers, away from the area of lymphocytic attack; the nuclear expression of p53 observed was predominantly negative, since 81.4 % of the samples showed a positivity average lower than 4% and whereas the remaining 18.60% showed a weak presence of p53 in the band-like subepithelial infiltrate, this could be an indication of apoptotic activity in these parts of the tissue, as well as the presence of cell damage in the basal layer, and the weak expression could be explained by the rapid proteolysis of this protein, as well as the fact that it is only involved during the G1 phase of the cell cycle (Fig. 1. B).
The average cell proliferation index at the nuclear level for Ki-67 was 12.40%, and proliferative cells were observed mainly in the basal layer of the epithelium (Fig. 1. C), while MCM3 showed moderate to strong nuclear positivity in almost all cases with average cell proliferation of 33.36% also in basal keratinocytes (Fig. 1. D); in turn, positivity at the nuclear level for histone H3 (H3) was predominantly weak, followed by moderate positivity, with a final average cell proliferation index of 26.76% and also found in the basal layer (Fig. 1. E).
CD-138 was found to be mostly strongly positive, with 95.35% at the level of the cell membrane throughout the epithelium, without negative expression in any of the cases (Fig. 1. F).
Lesions showed an active proliferation index in the keratinocytes of the basal and suprabasal layers, a slight increase in proapoptotic proteins in the basal layer and subepithelial inflammatory infiltrate (Table 1).
Discussion
BAX is considered an inducer of apoptosis and is a member of the BCL-2 family17. This family is formed by a network of protein-protein interactions that regulate apoptosis through permeabilization of the mitochondrial outer membrane. BAX is translocated to the mitochondria, where it is activated and integrated into the membrane, which causes a series of proteins considered apoptotic factors to be released to the cytoplasm (Fig. 2), (Cytochrome c, Smac/Diablo, Omi/HtrA2, Endonuclease G, apoptosis-inducing factor)18. High levels of expression are associated with a favorable prognosis in several types of cancer19-20.
The Bcl-2 oncogene encodes a protein which blocks a specific passage in the apoptotic pathways (Fig. 2). Abnormal expression of BAX, generally overexpressed in tumor cells, contributes to the spread of cell damage and the reduction of apoptosis; by enabling cell survival, it makes the acquisition of mutations and malignant transformations easier21. Its function is basically to prevent the release of cytochrome c through the outer mitochondrial membrane given the formation of heterodimers with anti-apoptotic molecules such as BAX22. The increased expression of BCL-2 in a study conducted on laryngeal squamous-cell carcinoma was associated with an advanced disease stage23.
As for the expression of apoptosis markers (BAX and BCL-2) and the halting of the cell cycle (p53) in OLP, our results were very similar to those reported in the international literature. Bascones et al. found that in lesion areas, the expression was mild to moderate in the basal layer, and in the suprabasal layer decreased to a mild intensity or negativity24.
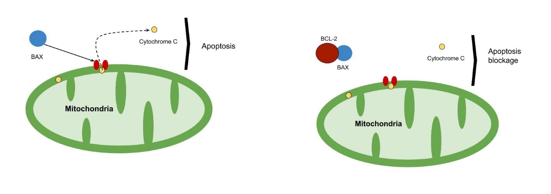
Fig. 2: When it binds to the membrane receptor, the pro-apoptotic biomarker BAX causes the release of cytochrome c from the mitochondria, which leads to cell death. When BCL-2 binds to this protein, this apoptosis pathway is blocked
In a study conducted on 30 cases of OLP, Shailaja et al. report a negative immunoexpression for BAX in 43.3% of the cases and moderate in 33.3%. While for BCL-2, immunoexpression was moderate in 36.7% of the cases and negative in 26.7%25. According to studies conducted by Bogdan et al. and Calenic et al.27, BCL-2 expression showed no significant differences when compared to the control group; unlike BAX immunoexpression, which was significantly higher than in the control group in both studies26-27. Moreover, the research conducted by Shailaja shows similar results regarding BAX expression compared to our study25.
The physiological function of p53 is to prevent the accumulation of genetic cell damage, whether through repair before cell or causing apoptosis. Alterations in p53 would, therefore, lead to uncontrolled cell growth28. It has been established that the functional damage of p53 is involved in the development and progression of oral epithelial dysplasia and oral squamous cell carcinoma29.
Several studies agree that p53 immunoexpression is significantly higher in OLP in the basal and suprabasal layers when compared to a control group (healthy oral mucosa)25-27,30-31.
Under normal conditions, p53 is found in low levels due to its rapid proteolysis27. Our results show weak expression of this protein at the level of the basal layer, suggesting an increase in cell apoptosis in the region.Ki-67 is a nuclear protein involved in the cell cycle, associated with cell proliferation and used as a cell proliferation marker to measure tumor cell growth28. This marker can be used for early detection of squamous-cell carcinoma, as it is expressed on phases G1, S, G2, M of the cell cycle32.
MCM3 is part of a set of proteins (minichromosome maintenance proteins) connected to DNA replication, and its increase plays a role in the malignant cell transformation process25. Several studies on MCM3 expression in lymphomas, leukemia, carcinoma of the uterine cervix, breast, kidney, stomach, lung, colon and melanoma, among others, have found that a high MCM3 expression is associated with a worse prognosis33.
Ki-67 is present during all active phases of the cell cycle, except G0 and early G1, whereas MCM3 is expressed in high levels during all phases of the cell cycle including early G1 and excluding G035.
In a study conducted by Rezazadeh et al. based on Ki-67 and MCM3 expression in oral squamous cell carcinoma, 96.4% of cases were positive for MCM3 and 78% for Ki-67, compared to the cases of normal mucosa where proliferative indices are low32. Cell proliferation measured by these two proteins in our study showed a weak immunoexpression of 92.68% for Ki-67 and a moderate one of 45.94% for MCM3, suggesting that it is elevated, which is reflected in the positive expression of both in OLP.
H3 is one of the five major histone proteins involved in the structure of chromatin of eukaryotic cells36. According to several studies, variations and modifications in the status of H3 play a role in long-term gene regulation37. Two genes, H3F3A and H3F3B, encode the H3 protein, but both are differentially regulated in carcinogenic processes, given that overexpression of H3F3A promotes cell invasion and cancer progression38-40. Finally, we found that H3 expression is weak to moderate, suggesting a mild to no alteration of this protein in connection with the pathogenesis of OLP or with its biological behavior.
Syndecan-1 (CD-138) is part of a family of receptors that take part in cell-cell and cell-matrix adhesion, and is localized in the basal and suprabasal level of cell layers38.
The expression of syndecan-1 is elevated during keratinocyte differentiation in the normal or hyperplastic epithelium and reduced in squamous cell carcinoma and premalignant cells; therefore, it may have a prognostic value in determining the clinical result of the lesion39. According to the study conducted by Manal et al., CD-138 expression is high and positive in normal stratified epithelium, but the expression is reduced in the cases of erosive OLP, and CD-138 immunoreactivity was associated with the lesions that were not malignized, which means this marker is considered to be potentially useful in determining the malignant potential of lesions31. In our study we found that CD-138 was conserved, which can be understood as a continuity in the cell-cell cohesion.
A considerable limitation of this study is that the expression of the proteins analyzed could not be countered or compared with the adjacent healthy tissue in all cases because some samples had insufficient tissue.
Conclusions
There appear to be pro-apoptotic mechanisms in the basal layer of the epithelium, which is supported by a higher expression of BAX compared to BCL-2; this speculation is reinforced by the fact that p53 was the only marker found in the subepithelial infiltrate.
The alteration of the proliferative mechanisms is associated with an increase in Ki-67, MCM3 and H3 biomarkers, suggesting a response of basal keratinocytes to lymphocyte-mediated immune damage toward the basal layer of the epithelium.
We suggest expanding this work in the future with a larger number of cases, comparing samples with healthy adjacent areas or normal oral mucosa of patients without OLP.
Referencias
1. Mc Comb D, Smith DC. A preliminary scanning electron microscopic study of root canals after endodontic procedures. J Endod. 1975; 1 (7): 238-242. [ Links ]
2. Baumgartner JC, Mader CL. A Scanning Electron Microscopic Evaluation of Four Root Canal Irrigation Regimens. J Endod. 1987; 13 (4): 147-157. [ Links ]
3. Mader Cl, Baumgartner JC, Peters DD. Scanning Electron Microscopic Investigation of the Smeared Layer on Root Canal Walls. J Endod. 1984; 10 (10): 477- 483. [ Links ]
4. Hülsmann M, Heckendorff M, Lennon A. Chelanting agents in root canal treatment: mode of action and indications for their use. Int Endod J. 2003; 36 (12): 810-830. [ Links ]
5. Orstavik D, Haapasalo M. Desinfection by endodontic irrigants and dressings of experimentally infected dentinal tubules. Endod Dent Traumatol. 1990; 6 (4): 142-149. [ Links ]
6. Kennedy WA, Walker WA, Gough RW. Smear layer removal effects on apical leakage. J Endod. 1986; 12 (1): 21-27. [ Links ]
7. Yamada RS, Armas A, Goldman M, Lin PS. A Scanning Electron Microscopic Comparison of a High Volume Final Flush with Several Irrigating Solutions: Part 3. J Endod 1983; 9 (4): 137-142. [ Links ]
8. Ostby NB. Chelation in root canal therapy. OdontolTidskr. 1957; 65: 1-11. [ Links ]
9. Von der Fehr FR, Ostby NB. Effect of EDTAC and sulfuric acid on root canal dentin. Oral Surg, Oral Med, Oral Pathol. 1963; 16 (2): 199-205. [ Links ]
10. Giardino L, Estrela C, Generali L, Mohammadi Z, Asgary S. The in vitro Effect of Irrigants with Low Surface Tension on Enterococcus faecalis. Iranian Endod J. 2015; 10 (3): 174-178. [ Links ]
11. Di Lenarda R, Cadenaro M, Sbaizero O. Effectiveness of 1 mol L-1 citric acid and 15% EDTA irrigation on smear layer removal. Int Endod J 2000; 33 (1): 46-52. [ Links ]
12. Haznedaroglu F. Efficacy of various concentrations of citric acid at different pH values for smear layer removal. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003; 96 (3): 340-344. [ Links ]
13. Wayman BE, Kopp WM, Pinero GL, Lazzari EP. Citric and lactic acids as root canal irrigants in vitro. J Endod. 1979; 5(9): 258-265. [ Links ]
14. Torabinejad M, Khademi AA, Babagoli J, Cho Y, Johnson WB, Bozhilov K et al.: A New Solution for the Removal of the Smear Layer. J Endod. 2003; 29 (3): 170-175. [ Links ]
15. Serper A, Calt S. The Demineralizing Effects of EDTA at Different Concentrations and pH. J Endod 2002; 28 (7): 501-502. [ Links ]
16. Seidberg BH, Schilder H. An evaluation of EDTA in endodontics. Oral Surg Oral Med Oral Pathol 1974; 37 (4): 609-620. [ Links ]
17. Schneider SW. A comparison of canal preparations in straight and curved root canals. Oral Surg Oral Med Oral Pathol . 1971; 32 (2): 271-275. [ Links ]
18. Schäfer E, Diez C, Hoppe W, Tepel J. Roentgenographic investigation of frequency and degree of canal curvatures in human permanent teeth. J Endod. 2002; 28(3): 211- 216. [ Links ]
19. Ricucci D, Siqueira JF. Fate of the Tissue in Lateral Canals and Apical Ramifications in Response to Pathologic Conditions and Treatment Procedures. J Endod. 2010; 36 (1): 1-15. [ Links ]
20. Khedmat S, Shokouhinejad N. Comparison of the Efficacy of Three Chelating Agents in Smear Layer Removal. J Endod. 2008; 34 (5):599-602. [ Links ]
21. Machado R, Garcia LDFR, da Silva Neto UX, Cruz Filho AMD, Silva RG, Vansan LP. Evaluation of 17% EDTA and 10% citric acid in smear layer removal and tubular dentin sealer penetration. Microsc Res Tech. 2018; 81 (3): 275-282 [ Links ]
22. Giardino L, Ambu E, Becce C, Rimondini L, Morro M. Surface Tension Comparison of Four Common Root Canal Irrigants and Two New Irrigants Containing Antibiotic. J Endod. 2006; 32(11): 1091-1093. [ Links ]
23. Goldberg F, Abramovich A. Analysis of the effect of EDTAC on the dentinal walls of the root canal. J Endod. 1977; 3 (3): 101-105. [ Links ]
24. Niu W. Yoshioka T. Kobayashi C. Suda H. A scanning electron microscopic study of dentinal erosion by final irrigation with EDTA and NaOCl solutions. Int Endod J. 2002; 35 (11): 934-939. [ Links ]
25. Calt S, Serper A. Time- Dependent Effects of EDTA on Dentin Structures. J Endod 2002: 28 (1): 17-19. [ Links ]
26. Baldasso FER, Roleto L, Silva VDD, Morgental RD, Kopper PMP. Effect of final irrigation protocols on microhardness reduction and erosion of root canal dentin. Braz Oral Res. 2017; 15: 31-40. [ Links ]
27. Turk T, Kaval, ME, Sen BH. Evaluation of the smear layer removal and erosive capacity of EDTA, boric acid, citric acid and desy clean solutions: an in vitro study. BMC Oral Health. 2015; 15: 104. [ Links ]
28. Marending M, Paqué F, Fischer J, Zehnder M. Impact of Irrigant Sequence on Mechanical Properties of Human Root Dentin. J Endod. 2007; 33 (11): 1325-1328. [ Links ]
29. Zhang K, Kim YK, Cadenaro M, Bryan TE, Sidow SJ, Loushine RJ, Ling JQ, Pashley DH, Tay FR. Effects of different exposure times and concentrations of sodium hypochlorite/ethylenediaminetetraacetic acid on the structural integrity of mineralized dentin. J Endod. 2010; 36 (1): 105-109 [ Links ]
30. Zhang K, Tay FR, Kim YK, Mitchell JK, Kim JR, Carrilho M, Pashley DH, Ling JQ. The effect of initial irrigation with two different sodium hypochlorite concentrations on the erosion of instrumented radicular dentin. Dent Mater. 2010; 26 (6): 514-523 [ Links ]
31. Tartari T, Bachmann L, Zancan RF, Vivan RR, Duarte MAH, Bramante CM. Analysis of the effects of several decalcifying agents alone and in combination with sodium hypochlorite on the chemical composition of dentine. Int Endod J. 2018; 51 Suppl 1: 42- 54. [ Links ]
32. Qian W, Shen Y, Haapasalo M. Quantitative Analysis of the Effect of Irrigant Solution Sequences on Dentin Erosion. J Endod. 2011; 37 (10): 1437-1441 [ Links ]
33. Haapasalo M, Qian W, Shen Y. Irrigation: beyond the smear layer. Endodontic Topics 2012; 27(1): 35-53 [ Links ]
35. Martinelli S, Strehl A, Mesa M. Estudio de la eficacia de diferentes soluciones de EDTA y ácido cítrico en la remoción del barro dentinario. Odontoestomatología. 2012; 14 (19): 52-63. [ Links ]
36. Bhasin M, Reinherz EL, Reche PA. Recognition and classification of histones using support vector machine. J Comput Biol2016; 13(1): 102-120 [ Links ]
37. Rosenfeld JA, Wang Z, Schones DE, Zhao K, DeSalle R, Zhang MQ. Determination of enriched histone modifications in non-genic portions of the human genome. BMC Genomics. 2009; 10: 143-154 [ Links ]
38. Carey DJ, Stahl RC, Tucker B, Bendt KA, Cizmeci-Smith G. Aggregation-induced association of syndecan - 1 white microfilaments mediated by the cytoplasmic domain. Exp Cell Res. 1994; 214: 12-21 [ Links ]
39. Soukka T, Pohjola J, Inki P, Happonen RP. Reduction of syndecan-1 expression is associated with dysplastic oral epithelium. J Oral Pathol Med. 2000; 29(7): 308-313 [ Links ]
40. Zink LM, Hake SB. Histone variants: nuclear function and disease. Curr Opin Genet Dev. 2016; 37: 82-89 [ Links ]
Received: May 06, 2019; Accepted: July 29, 2019










 texto en
texto en