Services on Demand
Journal
Article
Related links
Share
Ciencias Psicológicas
Print version ISSN 1688-4094On-line version ISSN 1688-4221
Cienc. Psicol. vol.13 no.2 Montevideo Dec. 2019 Epub Dec 01, 2019
https://doi.org/10.22235/cp.v13i2.1884
Original Articles
Comparison of light and deep sleep through heart rate variability
1 Facultad de Psicología, Universidad Nacional Autónoma México eriklms@ciencias.unam.mx, fayala@unam.mx, yoloyottl@gmail.com
2 Facultad de Ciencias de la Conducta, Universidad Autónoma del Estado de México c_gch@yahoo.com.mx
Keywords: cardiovascular activity; heart rate variability; NREM sleep; non-linear analysis
Palabras clave: actividad cardiovascular; variabilidad de la frecuencia cardiaca; sueño NMOR; análisis no lineal
Introduction
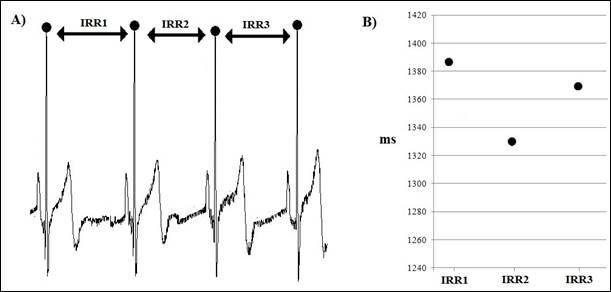
Psychophysiological states are dynamic processes that vary to individual adaptation to the demands from the internal and external environments. These processes can be reflected in different physiological variables, such as cardiac activity. The length of the heart inter-beat intervals (IBI) are not constant, as it changes according to the behavioral and physiological conditions of individuals. The study of the variations of these intervals is performed by means of the heart rate variability (HRV) analysis, which can provide information about psychophysiological states present in individuals under different circumstances (Billman, 2011). The HRV analysis can be addressed through different methods that are classified into time, frequency, and nonlinear domains (Task Force, 1996). It has been established that some methods provide information on the activity of the autonomic nervous system (ANS) under different physiological or behavioral conditions (Allen, Chambers, & Towers, 2007). In order to perform these analyses, it is required to obtain a measure of the IBI; by means of different methods using per example the electrocardiogram (EKG), whose intervals between R waves (RRI) are calculated (Figure 1A). Usually, RRIs are processed in two different ways; the first one consists in analyzing them directly, by means of their averages or standard deviations. The second way is to consider the RRIs as time series, each RRI being a value in the data sequence (Figure 1B). To represent them as time series these data are re-sampled at a fixed rate, usually using the interpolation method (Task Force, 1996). However, there are methods that process the data without re-sampling (Laguna, Moody, & Mark, 1998).
In the field of psychophysiology, HRV methods that provide measures of the level of activation of the ANS are widely used (Bernston et al., 1997; Laborde, Mosley, & Thayer, 2017). These measures have revealed differences between the Non-REM (NREM) and REM sleep (Lanfranchi, Pépin, & Somers, 2017; Mikoteit, De Witte, Holsboer-Trachsler, Hatzinger, Beck, & Pawlowski, 2019; Stein & Pu, 2012; Tobaldini, Nobili, Strada, Casali, Braghiroli, & Montano, 2013). Likewise, it has been also described the presence of HRV autonomic differences between light and deep NREM sleep (Ako, Kawara, & Uchida, 2003; Toscani, Gangemi, & Silipo, 1996; Trinder et al., 2001). However, NREM sleep division into two sleep kinds is not a standard procedure in the assessment of HRV.
There are other HRV measures that are also related to other phenomena, such as physiological variability, which has been proposed as a health indicator (Goldberger, 1997). Since NREM sleep is a heterogeneous phenomenon consisting of different phases, it is important to know which HRV measures also manifest this heterogeneity. Therefore, the objective of this study was to analyze and to compare the HRV during light and deep NREM sleep of healthy individuals.
Method
Participants
Thirty healthy volunteers participated in this study, but six were excluded for not meeting the experimental requirements, remaining as the final sample 24 participants, 14 women with 23.9 years (standard deviation SD 3.3) and 10 men with 26.2 years (SD 4.3).
The inclusion criteria were that participants did not have a diagnosis of chronic illness or psychiatric disorder, and the exclusion criteria were that participants smoke or use illegal drugs. The elimination criteria were the presence of some sleep disorder identified by means of polysomnography (PSG) or that the sleep percentages were not in the ranges considered normal. All participants were provided with information about the investigation procedure and subsequently signed an informed consent letter. In addition, the evaluation procedures were conducted in accordance with the basic principles set out in the Declaration of Helsinki.
Procedure
Polysomnographic records (PSG) were carried out during two consecutive nights, the first PSG was considered as night of adaptation and served to detect the presence of some indicator of sleep disorder. In the first PSG, the electroencephalogram (EEG) was recorded with contralateral references to the mastoids F3-M2, F4-M1, C3-M2, C4-M1, O1-M2 and O2-M1, the electrooculogram (EOG), the surface electromyography (EMG) of the chin and the right and left tibial muscles, EKG with derivation I, oronasal airflow recorded with thermistor, respiratory effort measured with thoracoabdominal bands and oxygen saturation recorded with pulse oximeter. In the second PSG the EEG, EOG, EMG of the chin and the EKG with derivation I were recorded. The PSGs were obtained with a Cadwell EasyII device (Kennewick, Washington, USA). The beginning of the recordings (lights off) was adjusted for each participant according to the usual time they used to go to sleep and allowed 8 hours to finish it. The classification of sleep stages was carried out according to the AASM method (Iber, Ancoli-Israel, Chesson, & Quan, 2007), light sleep corresponded to stage N2 and deep sleep corresponded to stage N3. For this study, only the data obtained during the second PSG of the participants were considered.
In the first and second NREM-REM cycles, 5-minute blocks of light and deep sleep EKG activity were selected. They were stable stages without electroencephalographic activation or movements. Cardiac activity samples that were 2 minutes before and after stage transitions were omitted. The EKG signal was exported in EDF format. QRSTool version 1.2.2 (Allen, Chambers, & Towers, 2007) was subsequently used to obtain the IBIs. Afterwards, CMet (Allen, Chambers, & Towers, 2007), CardioBatch (Brain-Body Center, University of Illinois, 2007) and Kubios HRV (Tarvainen, Niskanen, Lipponen, Ranta-aho, & Karjalainen, 2014) programs were used to calculate HRV measures.
Measures of HRV
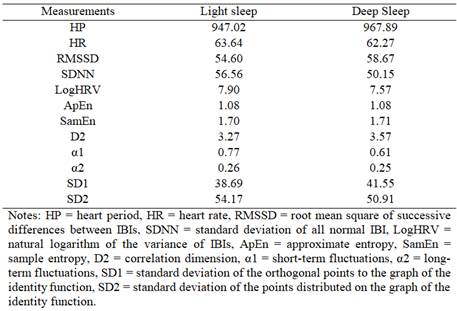
Within the time domain, the following measures were calculated: heart period (HP), heart rate (HR), root mean square of successive differences between IBIs (RMSSD), standard deviation of all normal IBI (SDNN) and the natural logarithm of the variance of the IBIs (LogHRV)
From the non-linear domain, the following measures were calculated: approximate entropy (ApEn), sample entropy (SamEn) and the correlation dimension (D2). In addition, from the detrended fluctuation analysis (DFA), short-term fluctuations were obtained by means of the slope α1 and long-term fluctuations by means of the slope α2. Finally, from Poincaré's graph, the standard deviation of the orthogonal points to the graph of the function identity (SD1) and the standard deviation of the distributed points on the graph of the function identity (SD2) were obtained.
Statistical analysis
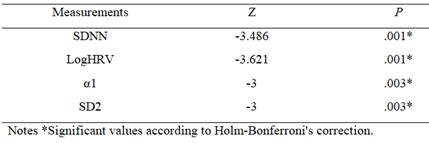
Due to the fact that not all the measurements obtained had a normal distribution, non-parametric tests were chosen. By means of the Wilcoxon signed-rank test, the data obtained from light and deep sleep were compared. In addition, since the number of tests involved multiple analyses and due to the lack of normal distribution it was applied the Holm-Bonferroni correction method (Holm, 1979) with a significance level of 0.05.
Results
A total of 435 blocks of 5 minutes of cardiac activity were collected, 162 corresponded to light sleep and 273 to deep sleep. Total sleep time observed during the 8 hr recording was 457.5 minutes (SD 37.7). The percentages of each sleep phase were for N1 of 6.9 (SD 2.7), N2 of 52.8 (SD 7), N3 of 19.7 (SD 5.8) and REM sleep of 20.6 (SD 3.9).
In relation to cardiac activity, when comparing the medians of each measure (Table 1), it was found that in the measures belonging to the non-linear domain higher values predominated during deep sleep (with the exception of α1 and SD2). Comparing the medians of each measure (Table 1), it was found that in the measures belonging to the non-linear domain higher values predominated during deep sleep (with the exception of α1 and SD2). On the other hand, three of the five time domain measures (HR, SDNN and logHRV) had higher values in light sleep. After the Holm-Bonferroni correction at the significance level, significant differences were found in 4 of the measures (Table 2), where median values were higher in light sleep.
Discussion
The measures in which significant differences were found correspond to indicators of general variability of cardiac functioning as described by other authors (Allen, Chambers, & Towers, 2007). In the case of the SDNN and logHRV measures, significant differences were observed between both types of sleep, with greater variability evident in light sleep. The above suggests that there are functional processes during light sleep reflexed as cardiac activity less stable, for example it has been reported that the presence of K complexes modifies blood pressure and cardiac activity (Silvani, 2008). However, the RMSSD measure, which also evaluates variability, showed higher values during deep sleep without reaching significant levels.
The procedure followed for the calculation of the RMSSD is different from that of SDNN and logHRV (Shaffer & Ginsberg, 2017), while the latter only differ in the way they represent the IBI values. This indicates that, of the three measures used within the time domain, there are at least two different ways to characterize the variability associated with cardiac functioning.
In the case of the non-linear domain measures related to variability, we found that SD2, from Poincaré's graph, showed significant differences between both sleep stages. This measure is also considered to reflect total variability and has been related to SDNN (Brennan, Palaniswami, & Kamen 2001). According to Goldberger (1997) another way to characterize cardiac variability is to evaluate its fractal characteristics such as self-similarity (when a part of the object is similar to the whole object). In this case, the DFA analysis quantifies fractal correlation properties of IBI series. In this study we found that the measure α1, which represents short-term fluctuations, was greater in light sleep. Penzel, Kantelhardt, Becker, Peter and Bunde (2003) using discriminant analysis described that the measures α1 and α2 were effective in differentiating sleep stages. These authors, in their analysis, included stage N1 within light sleep, in addition to adding REM sleep and wakefulness. The same authors observed that by including SDNN in the discriminant analysis, phase separation was more effective. In our study, we did not include phase N1 because few participants presented it for more than 5 minutes, which is common in this stage of NREM sleep (Carskadon & Dement, 2017). In addition, some authors propose that N1 is a transitional stage between wakefulness and sleep (Ogilvie, 2001).
Because the regulation of the cardiovascular system involves different mechanisms that interact with each other in a non-linear way (Ernst, 2017), the use of non-linear methods allow us to know characteristics of HRV that are not obtained with linear methods (Young & Benton, 2015). Entropy measures quantify the regularity and predictability of a system (Richman & Moorman, 2000), in this case no differences were found between both types of sleep with the two entropy measures analyzed (ApEn, SamEn). No significant differences were found in measure D2 either, this measure provides information on the minimum number of variables required to model the underlying system (Acharya, Joseph, Kannathal, Lim, & Suri, 2006).
With the measures used in this work it can be concluded that the differentiation between light and deep sleep depends on the type of analysis, based on the measures associated with cardiac variability, the division of NREM sleep could be justified in terms of cardiovascular activity. One factor that could contribute to this difference was the presence of compensatory rebound from deep sleep, although the sleep percentages of the second PSG were within normal parameters (Carskadon & Dement, 2017), in future studies it would be advisable to perform more than two PSG to compare HRV during light and deep sleep. Additionally, studies with larger samples are required for better characterization of light and deep sleep, as well as the inclusion of HRV measures that allow inference of ANS activity.
REFERENCES
Acharya, U. R., Joseph, K. P., Kannathal Lim, C. M. & Suri, J. S. (2006). Heart rate variability: a review. Medical & Biological & Engineering & Computing, 44 (12), 1031-1051. doi: 10.1007/s11517-006-0119-0 [ Links ]
Allen, J. J., Chambers, A. S. & Towers, D. N. (2007). The many metrics of cardiac chronotropy: A pragmatic primer and a brief comparison of metrics. Biological Psychology, 74 (2), 243-262. DOI: 10.1016/j.biopsycho.2006.08.005 [ Links ]
Ako, M., Kawara, T. & Uchida, S. (2003). Correlation between electroencephalography and heart rate variability during sleep. Psychiatry and Clinical Neurosciences, 57 (1), 59-65. doi: 10.1046/j.1440-1819.2003.01080.x [ Links ]
Berntson, G. G., Bigger, J. T., Eckberg, D. L., Grossman, P., Kaufmann, P. G., Malik, M.,… Van der Molen, M. (1997). Heart rate variability: Origins, methods, and interpretive caveats. Psychophysiology, 34 (6), 623-648. doi: https://doi.org/10.1111/j.1469-8986.1997.tb02140.x [ Links ]
Billman, G. E. (2011). Heart rate variability - a historical perspective. Frontiers in Physiology, 2, 1-13. doi: 10.3389/fphys.2011.00086 [ Links ]
Brennan, M., Palaniswami, M. & Kamen, P. (2001). Do existing measures of Poincare plot geometry reflect nonlinear features of heart rate variability? IEEE Transactions on Biomedical Engineering, 48 (11), 1342-1347. doi: 10.1109/10.959330 [ Links ]
Carskadon, M. A. & Dement, W. C. (2017). Normal human sleep: an overview. En M. H. Kryger, T. Roth y W. C. Dement (Eds.), Principles and practice of sleep medicine (pp. 15-24). Philadelphia PA: Elsevier. [ Links ]
Ernst, G. (2017). Heart rate variability - More than heart beats? Frontiers in Public Health, 5, 240. doi: 10.3389/fpubh.2017.00240 [ Links ]
Goldberger, A. L. (1997). Fractal variability versus pathologic periodicity: complexity loss and stereotypy in disease. Perspectives in Biology and Medicine, 40 (4), 543-561. doi: 10.1353/pbm.1997.0063 [ Links ]
Holm, S. (1979). A simple sequential rejective method procedure. Scandinavian Journal of Statistics, 6 (2), 65-70. doi: 10.2307/4615733 [ Links ]
Iber, C., Ancoli-Israel, S., Chesson, A. L. & Quan, S. F. (2007). The AASM manual for the scoring of sleep and associated events: rules, terminology and technical specifications. Westchester, IL: American Academy of Sleep Medicine. [ Links ]
Laborde, S., Mosley, E. & Thayer, J. (2017). Heart rate variability and cardiac vagal tone in psychophysiological research - Recommendations for experiment planning, data analysis, and data reporting. Frontiers in Psychology, 8, 1-18. doi: 10.3389/fpsyg.2017.00213 [ Links ]
Laguna, P., Moody, G. B. & Mark, R. G. (1998). Power spectral density of unevenly sampled data by least -square analysis: performance and application to heart rate signals. IEEE Transactions on Biomedical Engineering, 45 (6), 698-715. doi:10.1109/10.678605 [ Links ]
Lanfranchi, P. A., Pépin, J. & Somers, V. K. (2017). Cardiovascular physiology: autonomic control in health and in sleep disorders. En M. Kryger, T. Roth & W. C. Dement (Eds.), Principles and practice of sleep medicine (pp. 142-154). Philadelphia: Elsevier. [ Links ]
Mikoteit, T., De Witte, M., Holsboer-Trachsler, E., Hatzinger, M., Beck, J. & Pawlowski, M. (2019). Sleep stage related heart rate variability distinguishes between insomnia and normal sleep. Pharmacopsychiatry, 52 (2), 97. doi: 10.1055/s-0039-1679147 [ Links ]
Ogilvie, R. D. (2001). The process of falling asleep. Sleep Medicine Reviews, 5(3), 247-270. doi:10.1053/smrv.2001.0145 [ Links ]
Penzel, T., Kantelhardt, J. W., Becker, H. F., Peter, J. H. & Bunde, A. (2003). Detrended fluctuation analysis and spectral analysis of heart rate variability for sleep stage and sleep apnea identification. Computers in Cardiology, 30, 307-310. doi: 10.1109/CIC.2003.1291152 [ Links ]
Richman, J. & Moorman, J. (2000). Physiological time-series analysis using approximate entropy and sample entropy. American Journal of Physiology-Heart and Circulatory Physiology, 278 (6), H2039-2049. doi: 10.1152/ajpheart.2000.278.6.H2039 [ Links ]
Shaffer, F. & Ginsberg, J. P. (2017). An overview of heart rate variability metrics and norms. Frontiers in Public Health, 5, 218. doi: 10.3389/fpubh.2017.00258 [ Links ]
Silvani, A. (2008). Physiological sleep-dependent changes in arterial blood pressure: central autonomic commands and baroreflex control. Clinical and Experimental Pharmacology and Physiology, 35(9), 987-994. doi: 10.1111/j.1440-1681.2008.04985.x [ Links ]
Stein, P.K. & Pu, Y. (2012). Heart rate variability, sleep and sleep disorders. Sleep Medicine Reviews, 16 (1), 47-66. doi: 10.1016/j.smrv.2011.02.005 [ Links ]
Tarvainen, M. P., Niskanen, J. P., Lipponen, J. A., Ranta-aho, P. O. & Karjalainen, P. A. (2014). Kubios HRV - Heart rate variability analysis software. Computer Methods and Programs in Biomedicine, 113 (1), 210-220. doi: 10.1016/j.cmpb.2013.07.024 [ Links ]
Task Force of the European society of cardiology and the North American Society of pacing and Electrophysiology (1996). Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. European Heart Journal, 17, 354-381. doi: 10.1093/oxfordjournals.eurheartj.a014868 [ Links ]
Tobaldini, E., Nobili, L., Strada, S., Casali, K. R., Braghiroli, A. & Montano, N. (2013). Heart rate variability in normal and pathological sleep. Frontiers in Physiology, 4, 294. doi: 10.3389/fphys.2013.00294 [ Links ]
Toscani, L., Gangemi, P. F., & Silipo, R. (1996). Human heart rate variability and sleep stages. The Italian Journal of Neurological Sciences, 17(6), 437-439. doi: 10.1007/BF01997720 [ Links ]
Trinder, J., Kleiman, J., Carrington, M., Smith, S., Breen, S., Tan, N. & Kim, Y. (2001). Autonomic activity during human sleep as a function of time and sleep stage. Journal of Sleep Research, 10(4), 253-264. doi: 10.1046/j.1365-2869.2001.00263.x [ Links ]
Young, H. & Benton, D. (2015). We should be using nonlinear indices when relating heart-rate dynamics to cognition and mood. Scientific Reports, 5, 16619. doi: 10.1038/srep16619 [ Links ]
Note: Authors' participation: a) Conception and design of the work; b) Data acquisition; c) Analysis and interpretation of data; d) Writing of the manuscript; e) Critical review of the manuscript. E.L.M.S. ha contribuido en a,b,c,d,e; F.A.G. en a,c,d,e; K.A.P.P. en c,d,e; C.A.G.CF. en c,d,e.
Correspondence: Laboratorio de Neurociencias, Sótano del Edificio C, Facultad de Psicología, Av. Universidad 3004, Col. Copilco-Universidad 04510, Coyoacán, CDMX., México. E-mails: eriklms@ciencias.unam.mx; fayala@unam.mx; yoloyottl@gmail.com; c_gch@yahoo.com.mx
Received: November 21, 2018; Accepted: June 11, 2019










 text in
text in