Servicios Personalizados
Revista
Articulo
Links relacionados
Compartir
Ciencias Psicológicas
versión impresa ISSN 1688-4094versión On-line ISSN 1688-4221
Cienc. Psicol. vol.13 no.1 Montevideo jun. 2019
https://doi.org/10.22235/cp.v13i1.1809
Original Articles
Influence of depression symptoms on neuropsychological functions after stroke
1 Instituto de Psicologia, Universidade Federal do Rio Grande do Sul. Brasil piolidaniele@gmail.com
2Universidade do Vale do Rio dos Sinos. Brasil jaquecarvalhorodrigues@gmail.com
3Instituto de Psicologia, Universidade Federal do Rio Grande do Sul. Brasil jerusafsalles@gmail.com Correspondence: Daniele Pioli dos Santos, Instituto de Psicologia, Rua Ramiro Barcelos, nº 2600, sala 114, Bairro Santana, CEP 90035-003, Porto Alegre / RS / Brasil.
Abstract: The frequency of symptoms of depression and its influence on cognitive performance after stroke were analyzed. A total of 60 adults (M = 64.43, SD = 12.42 years) were evaluated from 102 to 283 days post-stroke (M = 140.65, SD = 28.01). Cognitive Screening to Stroke (Triagem Cognitiva nas Doenças Cerebrovasculares - TRIACOG), Rey-Osterrieth Complex Figure, Digits, Rey Verbal, FAS, Five Digit Test and Beck Depression Inventory II (BDI-II) were administered. Approximately 42% of the participants presented symptoms of post-stroke depression, with a higher frequency of Lack of energy, Change in sleeping pattern, Tiredness or fatigue, and Loss of interest in sex. There was a significative association among neuropsychological tasks and somatic items of BDI-II. Symptoms of depression and severity of the clinical neurological status explained between 22% and 33% of performance in memory, attention, and speed of processing. Symptoms of fatigue and post-stroke depression influenced negatively self-perception during the neuropsychological evaluation.
Key words: stroke; neuropsychological assessment; depression; neuropsychiatry; mental health
Palabras clave: accidente cerebrovascular; evaluación neuropsicológica; depresión; neuropsiquiatría; salud mental
Palavras-chave: acidente vascular cerebral; avaliação neuropsicológica; depressão; neuropsiquiatria; saúde mental
Introduction
Cerebrovascular Accident (CVA) is nowadays the first cause of death and impairment in Latin America and the second biggest cause of mortality and the third cause of disability worlwide (Avezum, Costa-Filho, Pieri, Martins, & Marin-Neto, 2015; Feigin, Norrving, & Mensah, 2017). The most common consequences of stroke are motor, cognitive and emotional alterations (Crichton et al., 2016; Eskes et al., 2015; Levine et al., 2015). Recurring post-stroke neuropsychological deficits are difficulties in memory, attention, language, perception, information processing speed, impairment on executive functions, praxis and arithmetic skills (Barker-Collo et al., 2016; Jokinen et al., 2015; Levine et al., 2015).
Emotional alterations as symptoms of depression and anxiety are among the most frequent psychiatric disorders after a stroke (Hackett, et al., 2014). Some of the most common neuropsychological alterations are impairment in reasoning, attention (keeping and sustaining), decision-making, difficulties in episodic memory, executive dysfunction and agitation or psychomotor delay (American Psychiatric Association (APA), 2014; Moore, Moseley, & Atkinson, 2010).
Post-stroke depression is estimated to occur in about 30 to 40% of individuals who had a stroke (Hackett et al., 2014; Robinson & Spaletta, 2010), affecting quality of life, mortality and functional capacity (Ayerbe et al., 2013). The main risk factors for post-stroke depression are the presence of depression prior to stroke (Ayerbe et al., 2013; Eskes et al., 2015), lack of family or social support, anxiety, severity of CVA neurological and clinical condition (Ayerbe et al., 2013) and cognitive difficulties such as memory, executive functions, attention and psychomotor processing speed (Kauhanen et al., 1999; Krishnadas & Cavanagh, 2012).
Studies about post-stroke mood disorders generally use the total scores of depression questionnaires to relate to patients’ cognitive profile. However, a deep analysis of the items on the scales could be useful to better understand the symptoms (among several ones that occur in depression) that significantly compromise neuropsychological functions.
Therefore, the general objective of this study is to analyze the influence of depression symptoms on the cognitive performance of adults three to six months after a stroke. The specific objectives were: a) to analyze the frequency of the items (indicators of depressive symptoms) on the Beck Depression Inventory - BDI-II (Gorenstein et al., 2011) in a sample of adults after a stroke, b) to identify the correlation of depression symptoms (items on the scale) with the results obtained in neuropsychological tasks and c) to verify how the variance of performance on neuropsychological instruments can be explained by depression symptoms.
For the first objective, the hypothesis is that patients with depression symptoms will show high frequency on BDI-II items regarding loss of pleasure, lack of energy, alterations on sleep, appetite and mood patterns, according to other studies (Cumming et al., 2010; de Man-van Ginkel et al., 2015; Spalletta, Ripa, & Caltagirone, 2005). Regarding the second objective, high total scores on BDI-II are expected, related to a low performance in the attention, memory, processing speed and executive functions in neuropsychological tasks (Kauhanen et al., 1999; Krishnadas & Cavanagh, 2012). Finally, depression is estimated to have a negative influence on cognitive performance in episodic memory, attention and processing speed, explaining around 40% of neuropsychological variance (Barker-Collo et al., 2016; Robinson & Spalletta, 2010).
Method
Participants
Sixty adults participated in this study (M = 64.43; SD = 12.42 years old) assessed 102 to 283 days after a stroke (M = 140.65; SD = 28.01 days), 58 right-handed and four left-handed patients. The necessary sample to identify a difference of dz = .15, considering the probability α = .10, and statistical power 1 - β = .90, is 45 subjects calculated by the G * Power software (Faul et al., 2007). Participants had at least three years of formal education, no record of learning difficulty, psychiatric diseases, dementia or other neurological injuries, such as head injury and brain tumor (according to neuroimaging tests). The sample did not include patients reporting use of illicit drugs. Patients showed heterogeneity in the severity of neurological clinical condition and in specific locations of brain injury (Table 1).
Procedures and Instruments
The study was approved by research ethics committees in a public university (CAAE 33603214.5.0000.5334) and a public hospital in the South of Brazil (CAAE 40624415.2.3001.5327). Patients were asked to participate three months after the stroke, as recommended by the medical team and medical record analyses. After being informed about the research procedures, the participants or their guardians signed the informed consent, authorizing the use of results for scientific purposes.
Participants answered a questionnaire of sociodemographic data and health conditions (whether they were on any kind of rehabilitation and what treatments or medications were being administered). A neuropsychological assessment questionnaire was applied afterwards. All instruments applied psychometric properties validated to Brazil:
a) Cognitive Screening for Cerebrovascular Diseases (TRIACOG; Rodrigues, 2017): in approximately 25 minutes it assesses the following domains: orientation, verbal episodic and semantic memory, constructive praxis, verbal fluency, auditory attention and work memory, semantic memory, ideomotor praxis, reading, inference processing, naming, oral and written comprehension, dictation writing, repetition, dyscalculia, information processing speed, executive functions (alternation and inhibition) and episodic visual memory.
b) Beck Depression Inventory - BDI-II (Gorenstein et al., 2011): self-report questionnaire of 21 items used to measure the intensity of depressive symptoms. BDI-II scores range from zero to 63 points: 0-9 indicates no depression; 10-18- suggests mild to moderate depression; 10-29 moderate to severe depression; and 30-63 severe depression. BDI-II was fully read to all participants to ensure its understanding.
c) Rey Complex Figure Test - figure A (Oliveira & Rigoni, 2010): participants are asked to copy a semi-complex figure and three minutes later copy it from memory. It assesses visual perception, constructive praxis, planning capacity and episodic visual memory (immediate).
d) Digits (Wechsler Intelligence Adult Scale - WAIS-III; Nascimento, 2004): sequence of digits that must be recalled in direct order and subsequently in the reverse order stated by the examiner. It is a measure for auditive attention and work memory (operational).
e) Rey Auditory Verbal Learning Test - (Fichman et al., 2010; Paula et al., 2012): a list of words called repeatedly and a list of memory interference, which assesses verbal learning, long and short term verbal episodic memory.
f) Verbal fluency with a phoneme criterion (Esteves et al., 2015; Opasso, Barreto, & Ortiz, 2016): The participant is asked to call words beginning with the letters F, A and S during a minute each. It assesses language, attention, flexibility, inhibition, long-term memory and processong speed.
g) Five Digit Test - FDT (Sedó, M., de Paula, J. J., & Malloy-Diniz, L. F., 2015): reading of numbers and symbols divided into four parts. It assesses executive component functions, such as visual attention, inhibition, flexibility and processing speed.
During hospitalization, patients were assessed by physicians and neurology residents through the National Institute of Health Stroke Scale- NIHSS (Brott et al., 1989), which analyzes post-stroke motor and cognitive aspects, as well as the Rankin Scale (Caneda et al., 2006; de Haan et al., 1995), which assesses functional independence after a stroke. These data were recorded to be related to patients’ neurological profile by the use of the hospital discharge score.
Data Analysis
The study initially analyzed the frequencies of neurological and sociodemographic profiles, as well as BDI-II data provided by the participants. Raw scores in verbal fluency (FAS) (Machado et al., 2009; Opasso et al., 2016; Tombaugh, 1999), Rey Verbal (Magalhães & Hamdan, 2010) and TRIACOG (Rodrigues, 2017) were transformed into a Z score, according to the tests’ normative scores. Rey Complex Figure (Oliveira & Rigoni, 2010) and FDT (Sedó et al., 2015) had their scores changed into percentile, according to their norms. Finally, the scoring of Digits (Nascimento, 2004) was adapted to weighted score, adjusting results to age, according to their norms.
The correlation between BDI-II total score and socio-demographic data (age and education) and the neurological clinical status (time after stroke, NIHSS and Rankin) in order to verify whether there was any confounding variable in the sample. Right after that, correlations between BDI-II items and performance in neuropsychological tasks were carried out through Pearson correlations (p ≤ .05).
The tasks that presented significant correlation with BDI-II and significance level between .10 and .20 were included in a multiple regression model through the Enter method in order to explore how depressive symptoms can explain the variance in performance in the neuropsychological tasks. Neurological assessment scores (NIHSS and Rankin) were also added to the models once they are important variables influencing neurological performance besides depression symptoms (Barker-Collo et al., 2016). Therefore, independent variables are the BDI-II, NIHSS and Ranking total scores, and dependente variables are the results of neuropsychological tests (in percentile, Z score and weighted score). Data analyses were performed by SPSS Statistics version 20.
Results
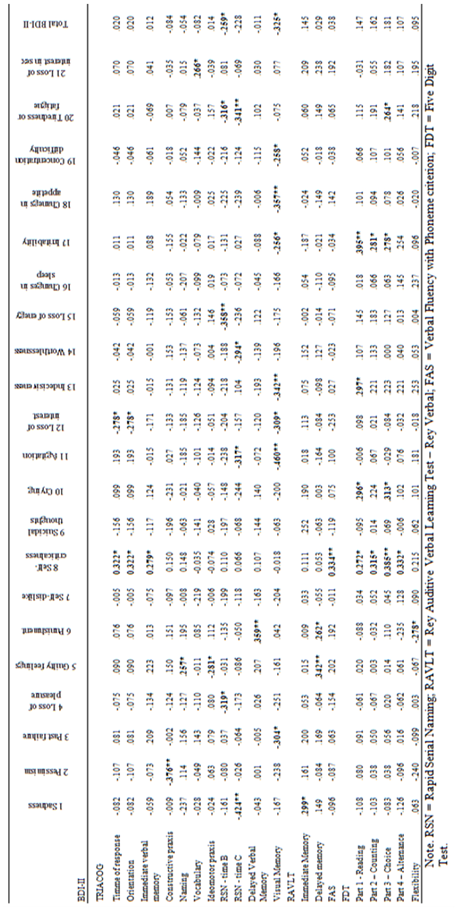
According to the BDI-II classification (Gorenstein et al., 2011), 58.3% of participants showed zero or minimum symptoms of depression, 16.7% mild symptoms, 10% showed moderate symptoms and 15% of the sample showed severe symptoms. Percentages related to each item of the instrument are presented in Table 2.
Participants showed higher frequency (percentage) of scores in the following items: Loss of energy, Changes in sleep, Tiredness or fatigue and Loss of interest in sex after the stroke, according to BDI-II. Higher frequency of maximum scoring was observed (severe symptom - score 3) in the items Punishment feelings, Agitation, Loss of interest, Changes in sleep, Tiredness or fatigue and Loss of interest in sex (Table 2).
The total BDI-II score did not seem to be related to education, severity of clinical neurological status and functional dependence variables. Regarding neuropsychological variables, Table 3 describes only those correlated with some of the BDI-II items.
Total TRIACOG seemed to be influenced by the severity of neurological status according to the NIHSS scale (R2 = .33; β = -.40; p = .010), as well as depression symptoms according to BDI-II (R2 = .33; β = -.28; p = .031). BDI-II scores (R2 = .22; β = -.37; p = .007) and NIHSS scores (R2 = .22; β = -.38; p = .022) showed influence of reading time in the Rapid Serial Naming (part B). BDI-II was also important to explain the performance in the Visual Memory of TRIACOG (R2 = .24; β = -.31; p = .019), as well as NIHSS (R2 = .24; β = -.37; p = .023).
Discussion
The distribution of participants among the categories of performance on BDI-II corroborates the finding that 40% of the adults who experienced stroke show depression symptoms at a certain level (Robinson & Spalletta, 2010). In this study, participants were assessed between three to six months after the stroke, providing na indicator of this specific post-stroke period.
Regarding the first objective of this study, it has confirmed the hypothesis of high prevalence of the following BDI-II items: Loss of energy, Tiredness or Fatigue, Changes in sleep and Loss of interest in sex. The prevalence of these items can be attributed to the overlap with symptoms of post-stroke fatigue (Chen et al., 2015; Eskes et al., 2015). Many patients may present motor alterations after a stroke, which compromises their mobility and causes more fatigue. The change in the spouse’s role, who often becomes the carer for that patient with limitations after the stroke, may also be related to the prevalence of the item Loss of interest in sex, as well as the one which is related to aging (Monteiro et al., 2012; Thompson & Ryan, 2009).
The results of correlations between BDI-II items and neuropsychological tests corroborate the hypothesis regarding the second objective. There was a connection between lower scores in the tasks assessing memory, attention and executive functions and higher levels of depression symptoms (Barker-Collo et al., 2016; Jokinen et al., 2015). The neuropsychological assessment showed that more critical self-perception led patients to check their performance in the tests more rigidly, which might have influenced the positive correlation between time of response on TRIACOG and the Self-criticalness item of BDI-II. On the other hand, participants with higher Self-criticalness showed better performance in the Orientation task from TRIACOG. Individuals who are more critical towards their cognitive difficulties are believed to develop coping techniques for their deficits, which helped them to answer this task.
The negative perception of the post-stroke status might also have influenced the participants’ performance in tasks that involve different memory processes. Derogatory judgment during the assessment, even when the skills tested were preserved, seems to justify the correlations observed between Self-criticalness and Phonemic verbal fluency, Punishment feelings and Delayed verbal memory of TRIACOG, and between Guilty feelings and Delayed verbal memory of Rey Verbal, corroborating the correlation between subjective memory complaints and depression symptoms in a sample of Brazilian elders with no difficulties in neuropsychological tests found by Guerreiro et al. (2006).
During the assessments, participants frequently reported stressful situations when readapting to daily life routines after leaving the hospital. It corroborates the hypothesis that the correlations with psychological items of BDI-II, such as Past failure, Punishment and Guilty feelings may indicate a negative self-evaluation, possibly showing patients’ levels of self-esteem according to Santos et al. (2012).
Experiencing limitations during the assessment was a reason for the difficulty to carry out the constructive praxis subtest (in which the individual is asked to copy a semi-complex figure). Participants were demotivated to complete the figure when they showed a negative perception of their global performance, corroborating the correlation between this sub-test and the Pessimism item. Still, the understanding of the limitations after the stroke associated with high critical judgment may have affected the correlations between the Visual memory sub-test of TRIACOG and the Past failure, Agitation, Loss of interest, Indecisivess, Changes in appetite items and the total BDI-II scores, showing an association between negative self-perception, depression symptoms and lowered neuropsychological performance. At the moment of assessment, some participants showed avoidance and difficulty to engage in the tasks proposed. Tiredness or fatigue symtoms might have harmed patients’ performance (Chen et al., 2015; Eskes et al., 2015).
Depression symptoms and the severity of clinical neurological status explained altogether 33% of the variance in performance and neuropsychological assessment after a stroke on TRIACOG and influenced mainly the visual memory, inhibition and processing speed functions, corroborating the third hypothesis of this study. Barker-Collo et al. (2016) reported that depression has significant influence on, among other processes, attention and visual memory after the stroke, and that depressive symptoms associated with NIHSS (evaluation scale of the severity of clinical neurological status) were predictive for negative impact of neuropsychological deficits in participants’ functionality 12 months after the stroke.
The final result of TRIACOG was affected by depressive symptoms. However, the NIHSS score had a little more influence on the overall performance of the neuropsychological screening instrument. This might have occurred due to the fact that other neuropsychological processes assessed in this study did not seem significantly correlated with the BDI-II items in the sample and, therefore, do not help explain the variance of performance in the tests.
As previously discussed, the correlations with somatic items of BDI-II can occur due to demotivation and difficulty of engagement, as well as tendency to fatigue, which might explain the influence of depression symptoms in variance of performance in Rapid Serial Naming tasks (part B), which involves inhibition and information processing speed (executive functions) and Episodic visual memory of TRIACOG.
BDI-II did not correlate with demographic variables, neurological clinical status or functional dependence. Contrary to common sense, the participants who require the most care did not seem to have more depressive symptoms. It is important to notice that, excluding the most severe cases and those with physical or cognitive impairment to carry out the neuropsychological assessment may not represent all the patients’ profiles, once studies point to the severity of stroke and functionality after the event as predictors of post-stroke depression (Ayerbe et al., 2013; Robinson & Spalletta, 2010).
Another limitation of this study was the difficulty to control the occurrence of depression prior to stroke and the possible use of anti-depressants. Nevertheless, this study showed the connection between the current depression symptoms and neuropsychological measures. Further research might analyze the connection between these variables, the use of medication and previous clinical status. Finally, further studies can also explore the influence of other neuropsychiatric conditions concurrent to depression (anxiety, for instance) to understand their impact on neuropsychological déficits after a stroke.
Conclusion
This study assessed the influence of depression symptoms in neuropsychological functions after a stroke. The assessment of the association between the severity of the neurological clinical status and the neuropsychological performance was not predicted in the objectives of this study, but this association was also significant in the statistical analyses.
Depressive symptoms and neurological clinical status explained 22 to 33% of the variance in participants’ performance in the neuropsychological assessment, corroborating other studies in this area. Analyses showed higher prevalence of somatic symptoms in BDI-II, indicating some overlap of depressive symptoms and fatigue after a stroke that were relevant for the engagement and neuropsychological performance in the tests proposed. Visual memory, attention and processing speed were the neuropsychological functions related to depression symptoms in the regressions. However, considering the correlations, there are other neuropsychological processes also associated with port-stoke depression.
This study analyzes in detal the items that compose a depression scale (BDI-II) and the associations with several neuropsychological functions. Moreover, it provides wide understanding on patients’ neuropsychiatric profiles, detailing the most frequent port-stroke depression symptoms and neuropsychological deficits. These data can help specific interventions in order to improve patients’ quality of life.
This study can help qualify care and attention towards post-stroke patients in a multi-disciplinary approach, as well as guide the implementation of public policies for this population. The most frequent depression symptoms can be worked in therapy groups, guidance to family members and carers in order to approach these subjective aspects of the illness which influence prognosis, motivation for treatment and independence for daily routine, for instance.
REFERENCES
American Psychiatric Association. (2014). DSM-5: Manual diagnóstico e estatístico de transtornos mentais. Porto Alegre: Artmed. [ Links ]
Avezum, Á., Costa-Filho, F. F., Pieri, A., Martins, S. O., & Marin-Neto, J. A. (2015). Stroke in Latin America: Burden of disease and opportunities for prevention. Global Heart, 10(4), 323-331. doi: 10.1016/j.gheart.2014.01.006 [ Links ]
Ayerbe, L., Ayis, S., Wolfe, C. D. A., & Rudd, A. G. (2013). Natural history, predictors and outcomes of depression after stroke: Systematic review and meta-analysis. The British Journal of Psychiatry, 202(1), 14-21. doi: 10.1192/bjp.bp.111.107664 [ Links ]
Barker-Collo, S., Krishnamurthi, R., Feigin, V., Jones, A., Theadom, A., Barber, P. A., … Bennett, D. (2016). Neuropsychological outcome and its predictors across the first year after ischaemic stroke. Brain Impairment, 17(02), 111-122. doi:10.1017/BrImp.2016.17 [ Links ]
Brott, T., Adams, H. P., Olinger, C. P., Marler, J. R., Barsan, W. G., Biller, J., … Hertzberg, V. (1989). Measurements of acute cerebral infarction: A clinical examination scale. Stroke, 20(7), 864-70. [ Links ]
Caneda, M. A. G., Fernandes, J. G., De Almeida, A. G., & Mugnol, F. E. (2006). Confiabilidade de escalas de comprometimento neurológico em pacientes com acidente vascular cerebral. Arquivos de Neuro-Psiquiatria, 64(3A), 690-697. doi: 10.1590/S0004-282X2006000400034 [ Links ]
Chen, Y. K.,Qu, J.-F., Xiao, W. M., Li, W. Y.,Weng, H. Y., Li, W., … Xiang, Y. T. (2015). Poststroke fatigue: Risk factors and its effect on functional status and health-related quality of life. International Journal of Stroke , 10(4), 506-512. doi: 10.1111/ijs.12409. [ Links ]
Crichton, S. L., Bray, B. D., McKevitt, C., Rudd, A. G., & Wolfe, C. D. A. (2016). Patient outcomes up to 15 years after stroke: Survival, disability, quality of life, cognition and mental health. Journal of Neurology, Neurosurgery, and Psychiatry, 87(10), 1091-1098. doi: 10.1136/jnnp-2016-313361 [ Links ]
Cumming, T. B., Churilov, L., Skoog, I.,Blomstrand, C., & Linden, T. (2010). Little evidence for different phenomenology in poststroke depression. Acta Psychiatrica Scandinavica, 121(6), 424-430. doi: 10.1111/j.1600-0447.2010.01558.x [ Links ]
de Haan, R., Limburg, M., Bossuyt, P., van der Meulen, J., & Aaronson, N. (1995). The clinical meaning of Rankin ‘Handicap’ grades after stroke. Stroke, 26, 2027-2030. doi: 10.1161/01.STR.26.11.2027 [ Links ]
de Man-van Ginkel, J. M., Hafsteinsdóttir, T. B., Lindeman, E., Geerlings, M. I., Grobbee, D. E., & Schuurmans, M. J. (2015). Clinical manifestation of depression after stroke: Is it different from depression in other patient populations? PloSOne, 10(12), 1-12. doi: 10.1371/journal.pone.0144450 [ Links ]
Eskes, G. A., Lanctôt, K. L., Herrmann, N., Lindsay, P., Bayley, M., Bouvier, L., … Heart Stroke Foundation Canada Canadian Stroke Best Practices Committees. (2015). Canadian stroke best practice recommendations: Mood, cognition and fatigue following stroke practice guidelines, update 2015. International Journal of Stroke , 10(7), 1130-1140. doi: 10.1111/ijs.12557 [ Links ]
Esteves, C. S., Oliveira, C. R., Moret-Tatay, C., Navarro-Pardo, E., Carli, G. A. D., Silva, I. G., …Argimon, I. I. de L. (2015). Phonemic and semantic verbal fluency tasks: Normative data for elderly Brazilians. Psicologia: Reflexão e Crítica, 28(2), 350-355. doi: 10.1590/1678-7153.201528215 [ Links ]
Faul, F., Erdfelder, E., Lang, A.-G., & Buchner, A. (2007). G*Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behavior Research Methods, 39(2), 175-91. Retrieved from http://www.ncbi.nlm.nih.gov/pubmed/17695343 [ Links ]
Feigin, V. L., Norrving, B., & Mensah, G. A. (2017). Global burden of stroke. Circulation Research, 120(3), 439-448. doi: 10.1161/CIRCRESAHA [ Links ]
Fichman, H. C., Teresa Dias, L. B., Fernandes, C. S., Lourenço, R., Caramelli, P., & Nitrini, R. (2010). Normative data and construct validity of the Rey Auditory Verbal Learning Test in a Brazilian elderly population. Psychology & Neuroscience, 3(1), 79-84. doi: 10.3922/j.psns.2010.1.010 [ Links ]
Gorenstein, C., Pang, W. Y., Argimon, I. L., & Werlang, B. S. G. (2011). BDI-II - Inventário de depressão de Beck. Porto Alegre: Casa do Psicólogo. [ Links ]
Guerreiro, T. C., Veras, R., Motta, L. B., Veronesi. A. S, & Schmidt., S (2006). Queixa de memória e disfunção objetiva de memória em idosos que ingressam na Oficina da Memória na UnATI/UERJ. Revista Brasileira de Geriatria e Gerontologia, 9(1), 7-20. [ Links ]
Hackett, M. L., Köhler, S., O'Brien, J. T., & Mead, G. E. (2014). Neuropsychiatric outcomes of stroke. The Lancet Neurology, 13(5), 525-34. doi: 10.1016/S1474-4422(14)70016-X [ Links ]
Jokinen, H., Melkas, S., Ylikoski, R., Pohjasvaara, T., Kaste, M., Erkinjuntti, T., & Hietanen, M. (2015). Post-stroke cognitive impairment is common even after successful clinical recovery. European Journal of Neurology, 22(9), 1288-1294. doi: 10.1111/ene.12743 [ Links ]
Kauhanen, M., Korpelainen, J.T., Hiltunen, P., Brusin, E., Mononen, H., Määttä, R., … Myllylä, V. V. (1999). Poststroke depression correlates with cognitive impairment and neurological deficits. Stroke, 30(9), 1875-1880. [ Links ]
Krishnadas, R., & Cavanagh, J. (2012). Depression: An inflammatory illness? Journal of Neurology, Neurosurgery, and Psychiatry , 83(5), 495-502. doi: 10.1136/jnnp-2011-301779 [ Links ]
Levine, D. A., Galecki, A. T., Langa, K. M., Unverzagt, F. W., Kabeto, M. U., Giordani, B., & Wadley, V. G. (2015). Trajectory of cognitive decline after incident stroke. JAMA, 314(1), 41-51. doi: 10.1001/jama.2015.6968 [ Links ]
Machado, T. H., Fichman, H. C., Santos, E. L., Carvalho, V. A., Fialho, P. P., Koenig, A. M., … Caramelli, P. (2009). Normative data for healthy elderly on the phonemic verbal fluency task - FAS. Dementia & Neuropsychologia, 3(1), 55-60 [ Links ]
Magalhães, S. S., & Hamdan, A. C. (2010). The Rey Auditory Verbal Learning Test: Normative data for the Brazilian population and analysis of the influence of demographic variables. Psychology & Neuroscience, 3(1), 85-91. doi: 10.3922/j.psns.2010.1.011 [ Links ]
Monteiro, E. S., Zirpoli, M. O., Issa, I. Q., & Moreira, P. N.(2012). Disfunções sexuais em pacientes após acidente vascular cerebral. Revista Neurociências, 20(3), 462-467. [ Links ]
Moore, D. J., Moseley, S., & Atkinson, J. H. (2010). The influence of depression on cognition and daily functioning. In T. D. Marcotte & I. Grant (Eds.), Neuropsychology of everyday functioning (pp. 419-440). New York: The Guilford Press. [ Links ]
Nascimento, E. (2004). WAIS-III: Escala de Inteligência Wechsler para Adultos. Manual: Adaptação e padronização de uma amostra brasileira. São Paulo: Casa do Psicólogo. [ Links ]
Oliveira, M. S., & Rigoni, M. S. (2010). Figuras complexas de Rey - Manual. São Paulo: Casa do Psicólogo. [ Links ]
Opasso, P. R., Barreto, S. D. S., & Ortiz, K. Z. (2016). Phonemic verbal fluency task in adults with high-level literacy. Einstein, 14(3), 398-402. doi: 10.1590/S1679-45082016AO3629 [ Links ]
Paula, J. J., Melo, L. P. C., Nicolato, R., Moraes, E. N., Bicalho, M. A., Hamdan, A. C., & Malloy-Diniz, L. F. (2012). Fidedignidade e validade de construto do Teste de Aprendizagem Auditivo-Verbal de Rey em idosos brasileiros. Archives of Clinical Psychiatry, 39(1), 19-23. doi: 10.1590/S0101-60832012000100004 [ Links ]
Robinson, R. G., & Spalletta, G. (2010). Poststroke depression: A review. Canadian Journal of Psychiatry, 55(6), 341-349. doi: 10.1177/070674371005500602 [ Links ]
Rodrigues, J. C. (2017). Triagem cognitiva nas doenças cerebrovasculares: Processo de construção e propriedades psicométricas do instrumento TRIACOG. Tese de Doutorado não publicada. Programa de Pós-gradução em Psicologia. Universidade Federal do Rio Grande do Sul - UFRGS. [ Links ]
Santos, A. T., Leyendecker, D. D., Costa, A. L. S., & Souza-Talarico, J. N. (2012). Queixa subjetiva de comprometimento da memória em idosos saudáveis: Influência de sintomas depressivos, percepção de estresse e autoestima. Revista da Escola de Enfermagem da USP, 46(spe), 24-29. [ Links ]
Sedó, M., Paula, J. J., & Malloy-Diniz, L. F. (2015). FDT- Five Digit Test. Teste dos cinco dígitos. São Paulo: Hogrefe. [ Links ]
Spalletta, G., Ripa, A., & Caltagirone, C. (2005). Symptom profile of DSM-IV major and minor depressive disorders in first-ever stroke patients. The American Journal of Geriatric Psychiatry, 13(2), 108-115. doi: 10.1176/appi.ajgp.13.2.108 [ Links ]
Thompson, H. S., & Ryan, A. (2009). The impact of stroke consequences on spousal relationships from the perspective of the person with stroke. Journal of Clinical Nursing, 18(12), 1803-1811. doi: 10.1111/j.1365-2702.2008.02694.x [ Links ]
Tombaugh, T. (1999). Normative data stratified by age and education for two measures of Verbal Fluency FAS and Animal Naming. Archives of Clinical Neuropsychology, 14(2), 167-177. doi: 10.1016/S0887-6177(97)00095-4 [ Links ]
Note: Authors' participation: a) Conception and design of the work; b) Data acquisition; c) Analysis and interpretation of data; d) Writing of the manuscript; e) Critical review of the manuscript. D.P.S. ha contribuido en a,b,c,d; J.C.R. en a,b,c,d,e; J.F.S. en a,e.
Note: Acknowledgements: This study was made possible thanks to the financial support of Hospital de Clínicas de Porto Alegre (HCPA) Fundação de Amparo à Pesquisa do Rio Grande do Sul (FAPERGS) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).
Received: February 01, 2018; Revised: June 14, 2018; Accepted: February 20, 2019










 texto en
texto en