1. Introduction
The genus Erigeron (Asteraceae) includes herbaceous plants of ecological, medicinal, horticultural, and agronomic interest. Some species have been employed in traditional medicine for their anti-inflammatory and diuretic properties, while others, such as Erigeron breviscapus, contain bioactive compounds with antioxidant and potential neuroprotective effects 1)(2) . Conservation of these genetic resources in germplasm banks can support future pharmacological research, horticultural improvements, and ecological restoration efforts.
Several Erigeron species are also considered weedy, including hairy fleabane (E. bonariensis), sumatran fleabane (E. sumatrensis), and horseweed (E. canadensis). These weeds have shown remarkable adaptability, high seed output, widespread dispersal, and resistance to different herbicides 3)(4) . In Uruguay, E. bonariensis populations resistant to multiple herbicides (e.g., glyphosate and ALS inhibitors) have been reported 5)(6) . Likewise, E. sumatrensis populations resistant to multiple herbicide groups, including EPSPS-, ALS-, and PSI-inhibiting herbicides, as well as to 2,4-D, have been documented in Uruguay and South America 7)(8) 9. The evolution of herbicide resistance may reduce genetic diversity in natural populations, potentially leading to the loss of susceptible biotypes10. Germplasm banks can serve as reservoirs of genetic material from different biotypes, preserving diversity for research on resistance mechanisms, potential reintroduction, or restoration projects.
For orthodox seeds (those that tolerate drying and freezing), long-term storage is typically achieved by reducing seed moisture to about 7-6% and then storing at -20 °C in hermetically sealed containers 11)(12) . Standard seed storage theory indicates that each 1% decrease in seed moisture content or 5 °C reduction in temperature approximately doubles seed longevity12. Online databases indicate that all tested Erigeron species are likely orthodox13, but direct evidence for E. sumatrensis is lacking. Dormancy is also an important factor. Variable dormancy has been documented in Conyza species (synonymous with certain Erigeron species), influenced by temperature and light conditions 14)(15) 16. However, the exact nature of dormancy and the methods to overcome it remain unclear.
This study tested the hypothesis that short-duration drying at higher temperatures (e.g., 40 °C or 60 °C) could be as effective as the standard drying at 20 °C without affecting germination. Additionally, we aimed to confirm the orthodox behavior of E. sumatrensis seeds by evaluating the effect of a short-term (-20 °C for one month) freezing period on germination. The objectives were: (1) to evaluate the effect of different drying treatments on seed germination and (2) to assess the impact of freezing on germination of seed lots from different years (2019, 2020, and 2021).
2. Materials and Methods
Seeds of Erigeron sumatrensis were initially collected in 2019 from an agricultural field in Soriano, Uruguay (33°23'32.58"S, 57°59'4.08"W). Additional seed lots from 2020 and 2021 were produced under greenhouse conditions from the original collection at INIA La Estanzuela (34°20'23.72"S, 57°41'39.48"W), Colonia, Uruguay, during the indicated years. After harvest, seeds were cleaned manually and stored at approximately 7 °C until treatments were applied simultaneously to all seed lots, during the months of May and June, 2023, therefore using seeds of different age.
Three drying procedures were evaluated: (a) 20 days at 20 °C and 15% RH in a seed drying chamber (routine genebank protocol); (b) 10 days at 40 °C in a forced convection oven, and (c) 7 days at 60 °C in a forced convection oven. Moisture content was determined before and after drying using the oven method (105 °C for 17 hours)17. Following drying, seeds were subjected to different combinations of soaking and freezing (Table 1), as follows: Control: No drying, no freezing, no soaking; Soaking only: 24 h soaking in distilled water at 20 °C before germination; Drying at 20 °C + Soaking; Drying at 40 °C + Soaking; Drying at 60 °C + Soaking; Soaking + Freezing (-20 °C for 30 days); Drying at 20 °C + Freezing + Soaking; Drying at 40 °C + Freezing + Soaking; and Drying at 60 °C + Freezing + Soaking.
Table 1: Description of seed drying, storage time, soaking treatment, and humidity content measured in Erigeron sumatrensis seeds before germination
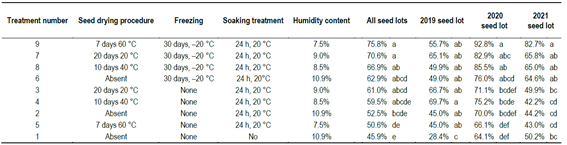
Germination least squared estimated means for the 14th day analyzed for all the combined and for each of the seed lots considering count dates as repeated measures. Values with the same letters within each column are not statistically different.
Due to the extremely small seed size and difficult handling, the one-month freezing period was selected based on the times used to determine conservation behavior for Erigeron species reported in the Seed Information Database13, which started from 17 days of freezing treatment (i.e., Erigeron acris, https://ser-sid.org/species/a3102298-df3e-40b6-9afb-04a4dfc58ae9). After treatments, four sub-samples of 50 seeds per lot were germinated in 90-mm Petri dishes on three layers of Whatman No. 1 filter paper moistened with 8 mL distilled water.
Germination took place in a chamber at 20 ± 2 °C, 90% RH, and a 12/12 h light/dark photoperiod, and germinated seeds were counted at 7 and 14 days, as found optimal in previous studies 18)(19) . Seeds were soaked into water to remove dormancy and facilitate germination as described by Kaspary and others10. A seed was considered germinated when all essential structures were present: root, stem, and leaves.
Statistical Analysis: Germination percentage was analyzed using a mixed model with a binomial distribution (SAS Proc GLIMMIX)20. Treatments were considered fixed effects and seed lots random. Germination count dates (7 and 14 days) were treated as repeated measures. Significant interactions were explored through contrasts (ESTIMATE and CONTRAST statements) and multiple comparisons using Dunnett’s test.
3. Results
A significant interaction between seed lot and treatment was found in germination at 14 days, leading to separate analyses by seed lot. Overall, soaking seeds for 24 h before germination increased average germination by about 6.6% across all lots. This effect was most pronounced for the older (2019) seed lot, where germination increased by 16.6%.
Drying at any of the tested conditions did not significantly reduce germination, indicating that even high-temperature drying (up to 60 °C) did not damage the seeds. Freezing at -20 °C for 30 days consistently increased germination, especially in newer (2020 and 2021) seed lots. The 2021 seed lot showed the strongest response to freezing, with germination increasing by approximately 26.2%. The combination of 60 °C drying followed by freezing yielded the highest germination rates. The older (2019) seed lot showed less improvement from these treatments, possibly due to natural aging and partial loss of viability over time.
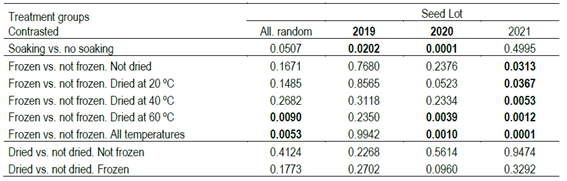
The results showed that a 24-hour soaking treatment caused a significant increase in Erigeron seeds germination. The soaking treatment of seeds before sowing increased mean germination by 6.6% at the 14th day (45.9% vs. 52.5%); the significance for this contrast is presented in Table 2, when compared to treatment without soaking (corresponding to the comparison between Treatments 1 and 2, Table 2). When analyzed by seed lot, the differences were 16.6% in the 2019 seed lot, 5.9% in the 2020 seed lot, and non-significant for the 2021 seed lot.
The germination percentage of the 2019 seed lot (Table 1) was less responsive to drying and storage treatments, which could be due to the fact that it was an older seed lot, in which any dormancy had already been lost.
The results also proved that germination increased 14.1% after freezing the seeds at -20 ºC (the average of treatments 3, 4, and 5 vs. 7, 8, and 9). This effect showed a variation of responses in the different seed lots, with no response for the 2019 seed lot, and a response that increases to 16.3% and to 26.2% for the 14th-day germination for the 2020 and 2021 seed lots, respectively; with a clear response for the 2021 seed lot across all drying temperatures (Table 1). The germination increase due to freezing was significant in all seed lots (Frozen vs. not frozen, all temperatures, Table 2), and also for the case of drying at 60 ºC and then freezing, for the 2020 and 2021 seed lots. The seed drying treatments across all drying temperatures and time combinations did not change germination significantly neither before nor after storage at -20 ºC (Table 2).
4. Discussion
The present study confirmed that Erigeron sumatrensis seeds can be classified as orthodox, demonstrating the feasibility of long-term conservation through standard drying and freezing protocols. Contrary to initial expectations, the 2021 seed lot did not consistently yield the highest germination rates despite its relatively recent harvest date. A common assumption in seed biology is that newer seed lots tend to have higher initial viability and higher levels of dormancy than older seed lots. While the 2021 seeds did show a strong response to freezing (particularly following high-temperature drying), the germination of 2021 seed lot was lower than the 2020 seed lot in all treatments.
Several factors may explain this outcome. First, seed maturation conditions in the greenhouse environment vary from year to year. Slight differences in temperature, humidity, day length, and nutritional status of the mother plants can influence seed quality, dormancy depth, and the ability to tolerate drying and freezing 1)(2) . Although the seed production environment was controlled, it is possible that environmental changes during seed formation led to variations in seed fill, chemical composition (e.g., lipid and protein content), or initial dormancy status that did not strictly correlate with harvest year. Another possibility is that the 2021 seed lot may have had a higher degree of primary dormancy at harvest compared to the 2020 lot, requiring more pronounced dormancy-breaking treatments. Dormancy expression can differ annually depending on maternal plant stress and seed maturation conditions 21)(22) 23. Thus, while the 2021 seeds responded positively to freezing and high-temperature drying, the starting point in terms of dormancy depth or viability may have prevented them from outperforming the 2020 seeds.
When viewed in the context of other studies on Erigeron and closely related Conyza species, our results align with previous reports that highlight environmental and maternal effects on seed germination behavior. Studies on Conyza bonariensis and C. canadensis -taxa closely related to E. sumatrensis- have demonstrated that germination is influenced not only by seed age but also by seed production environments and storage conditions 14)(15) . For example, Karlsson and Milberg15 found that after-ripening in C. canadensis and C. bonariensis was sensitive to storage environment; and Wu and others14 showed that seed persistence and emergence patterns in C. bonariensis depend on environmental cues. Similarly, variability in maternal effects and dormancy mechanisms has been documented in other Asteraceae species 7)(8) 9.
Within the Asteraceae family, species often exhibit variable dormancy mechanisms responsive to environmental signals, making direct comparisons between seed lots difficult. Research on other Asteraceae members has shown that germination can be strongly modulated by maternal environmental conditions and seed maturation timing 21)(22) 23. These studies support the idea that annual fluctuations in seed quality and dormancy depth can obscure a direct correlation between seed age and germination performance under standardized test conditions.
In summary, although one might assume that the most recently harvested seeds would have the highest germination potential, the complexity of dormancy mechanisms and maternal effects in Erigeron sumatrensis may have contributed to the observed patterns.
5. Conclusions
The results confirmed that Erigeron sumatrensis seeds behave as orthodox, indicating that long-term seed conservation with normal seed drying procedures is feasible. Both soaking treatments and drying at 60 °C in recently harvested seed lots may increase germination, perhaps by reducing seed dormancy. Conserving Erigeron seeds in a genebank will thus maintain the genus’s genetic variability for future exploration in academic research, and may also support their use in restoration, species reintroduction, or evaluation for other applications.